当前位置:
X-MOL 学术
›
Nat. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
C16-ceramide is a natural regulatory ligand of p53 in cellular stress response.
Nature Communications ( IF 14.7 ) Pub Date : 2018-10-08 , DOI: 10.1038/s41467-018-06650-y Baharan Fekry 1 , Kristen A Jeffries 1 , Amin Esmaeilniakooshkghazi 1 , Zdzislaw M Szulc 2, 3 , Kevin J Knagge 4 , David R Kirchner 4 , David A Horita 1 , Sergey A Krupenko 1, 5 , Natalia I Krupenko 1, 5
Nature Communications ( IF 14.7 ) Pub Date : 2018-10-08 , DOI: 10.1038/s41467-018-06650-y Baharan Fekry 1 , Kristen A Jeffries 1 , Amin Esmaeilniakooshkghazi 1 , Zdzislaw M Szulc 2, 3 , Kevin J Knagge 4 , David R Kirchner 4 , David A Horita 1 , Sergey A Krupenko 1, 5 , Natalia I Krupenko 1, 5
Affiliation
|
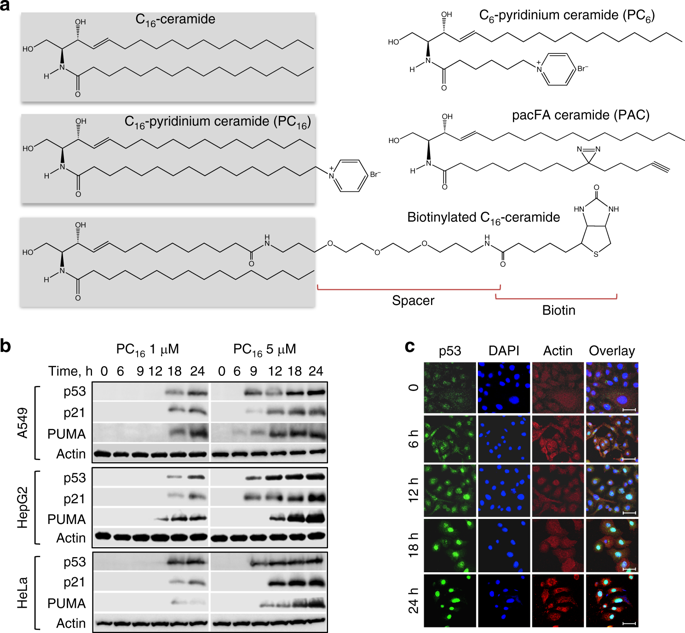
Ceramides are important participants of signal transduction, regulating fundamental cellular processes. Here we report the mechanism for activation of p53 tumor suppressor by C16-ceramide. C16-ceramide tightly binds within the p53 DNA-binding domain (Kd ~ 60 nM), in close vicinity to the Box V motif. This interaction is highly selective toward the ceramide acyl chain length with its C10 atom being proximal to Ser240 and Ser241. Ceramide binding stabilizes p53 and disrupts its complex with E3 ligase MDM2 leading to the p53 accumulation, nuclear translocation and activation of the downstream targets. This mechanism of p53 activation is fundamentally different from the canonical p53 regulation through protein-protein interactions or posttranslational modifications. The discovered mechanism is triggered by serum or folate deprivation implicating it in the cellular response to nutrient/metabolic stress. Our study establishes C16-ceramide as a natural small molecule activating p53 through the direct binding.
中文翻译:

C16-神经酰胺是细胞应激反应中p53的天然调节配体。
神经酰胺是信号转导的重要参与者,调节基本的细胞过程。在这里,我们报告了由C16-神经酰胺激活p53肿瘤抑制因子的机制。C16神经酰胺紧密结合在Box V基序附近的p53 DNA结合结构域(Kd〜60 nM)内。这种相互作用对神经酰胺酰基链长度具有高度选择性,其C10原子靠近Ser240和Ser241。神经酰胺结合可稳定p53并破坏其与E3连接酶MDM2的复合物,从而导致p53积累,核易位和下游靶标的活化。p53激活的这种机制从根本上不同于通过蛋白质-蛋白质相互作用或翻译后修饰进行的规范性p53调节。发现的机制是由血清或叶酸剥夺触发的,这涉及到对营养/代谢应激的细胞反应。我们的研究将C16-神经酰胺确定为通过直接结合激活p53的天然小分子。
更新日期:2018-10-08
中文翻译:

C16-神经酰胺是细胞应激反应中p53的天然调节配体。
神经酰胺是信号转导的重要参与者,调节基本的细胞过程。在这里,我们报告了由C16-神经酰胺激活p53肿瘤抑制因子的机制。C16神经酰胺紧密结合在Box V基序附近的p53 DNA结合结构域(Kd〜60 nM)内。这种相互作用对神经酰胺酰基链长度具有高度选择性,其C10原子靠近Ser240和Ser241。神经酰胺结合可稳定p53并破坏其与E3连接酶MDM2的复合物,从而导致p53积累,核易位和下游靶标的活化。p53激活的这种机制从根本上不同于通过蛋白质-蛋白质相互作用或翻译后修饰进行的规范性p53调节。发现的机制是由血清或叶酸剥夺触发的,这涉及到对营养/代谢应激的细胞反应。我们的研究将C16-神经酰胺确定为通过直接结合激活p53的天然小分子。






























 京公网安备 11010802027423号
京公网安备 11010802027423号