Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Multicomponent synthesis of tertiary alkylamines by photocatalytic olefin-hydroaminoalkylation
Nature ( IF 50.5 ) Pub Date : 2018-09-01 , DOI: 10.1038/s41586-018-0537-9 Aaron Trowbridge , Dominik Reich , Matthew J. Gaunt
Nature ( IF 50.5 ) Pub Date : 2018-09-01 , DOI: 10.1038/s41586-018-0537-9 Aaron Trowbridge , Dominik Reich , Matthew J. Gaunt
|
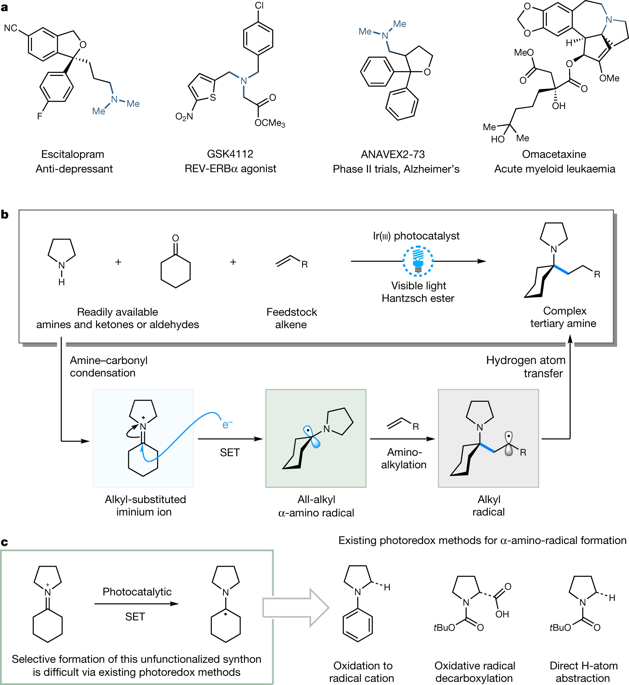
There is evidence to suggest that increasing the level of saturation (that is, the number of sp3-hybridized carbon atoms) of small molecules can increase their likelihood of success in the drug discovery pipeline1. Owing to their favourable physical properties, alkylamines have become ubiquitous among pharmaceutical agents, small-molecule biological probes and pre-clinical candidates2. Despite their importance, the synthesis of amines is still dominated by two methods: N-alkylation and carbonyl reductive amination3. Therefore, the increasing demand for saturated polar molecules in drug discovery has continued to drive the development of practical catalytic methods for the synthesis of complex alkylamines4–7. In particular, processes that transform accessible feedstocks into sp3-rich architectures provide a strategic advantage in the synthesis of complex alkylamines. Here we report a multicomponent, reductive photocatalytic technology that combines readily available dialkylamines, carbonyls and alkenes to build architecturally complex and functionally diverse tertiary alkylamines in a single step. This olefin-hydroaminoalkylation process involves a visible-light-mediated reduction of in-situ-generated iminium ions to selectively furnish previously inaccessible alkyl-substituted α-amino radicals, which subsequently react with alkenes to form C(sp3)–C(sp3) bonds. The operationally straightforward reaction exhibits broad functional-group tolerance, facilitates the synthesis of drug-like amines that are not readily accessible by other methods and is amenable to late-stage functionalization applications, making it of interest in areas such as pharmaceutical and agrochemical research.Complex tertiary alkylamines are prepared in one step from readily available amines, carbonyls and alkenes, via the visible-light-mediated reduction of in-situ-generated iminium ions to form alkyl-substituted α-amino radicals.
中文翻译:

光催化烯烃-加氢氨基烷基化多组分合成叔烷基胺
有证据表明,增加小分子的饱和度(即 sp3 杂化碳原子的数量)可以增加它们在药物发现管道中成功的可能性1。由于其良好的物理特性,烷基胺在药物制剂、小分子生物探针和临床前候选药物中无处不在。尽管它们很重要,胺的合成仍然由两种方法主导:N-烷基化和羰基还原胺化3。因此,药物发现中对饱和极性分子的需求不断增加,继续推动合成复杂烷基胺 4-7 的实用催化方法的发展。特别是,将可获取的原料转化为富含 sp3 的体系结构的工艺为复杂烷基胺的合成提供了战略优势。在这里,我们报告了一种多组分还原光催化技术,该技术结合了现成的二烷基胺、羰基化合物和烯烃,可在一步中构建结构复杂且功能多样的叔烷基胺。这种烯烃-氢氨基烷基化过程涉及可见光介导的原位生成亚胺离子的还原,以选择性地提供以前难以接近的烷基取代的 α-氨基自由基,这些自由基随后与烯烃反应形成 C(sp3)–C(sp3)债券。操作简单的反应表现出广泛的官能团耐受性,
更新日期:2018-09-01
中文翻译:

光催化烯烃-加氢氨基烷基化多组分合成叔烷基胺
有证据表明,增加小分子的饱和度(即 sp3 杂化碳原子的数量)可以增加它们在药物发现管道中成功的可能性1。由于其良好的物理特性,烷基胺在药物制剂、小分子生物探针和临床前候选药物中无处不在。尽管它们很重要,胺的合成仍然由两种方法主导:N-烷基化和羰基还原胺化3。因此,药物发现中对饱和极性分子的需求不断增加,继续推动合成复杂烷基胺 4-7 的实用催化方法的发展。特别是,将可获取的原料转化为富含 sp3 的体系结构的工艺为复杂烷基胺的合成提供了战略优势。在这里,我们报告了一种多组分还原光催化技术,该技术结合了现成的二烷基胺、羰基化合物和烯烃,可在一步中构建结构复杂且功能多样的叔烷基胺。这种烯烃-氢氨基烷基化过程涉及可见光介导的原位生成亚胺离子的还原,以选择性地提供以前难以接近的烷基取代的 α-氨基自由基,这些自由基随后与烯烃反应形成 C(sp3)–C(sp3)债券。操作简单的反应表现出广泛的官能团耐受性,































 京公网安备 11010802027423号
京公网安备 11010802027423号