Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2018-09-20 , DOI: 10.1016/j.bmc.2018.09.020 Tyler S Beyett 1 , Xinmin Gan 2 , Shannon M Reilly 3 , Andrew V Gomez 3 , Louise Chang 4 , John J G Tesmer 4 , Alan R Saltiel 3 , Hollis D Showalter 2
|
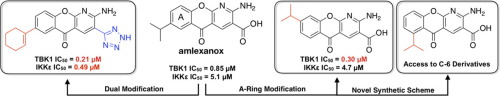
The non-canonical IκB kinases TANK-binding kinase 1 (TBK1) and inhibitor of nuclear factor kappa-B kinase ε (IKKε) play a key role in insulin-independent pathways that promote energy storage and block adaptive energy expenditure during obesity. Utilizing docking calculations and the x-ray structure of TBK1 bound to amlexanox, an inhibitor of these kinases with modest potency, a series of analogues was synthesized to develop a structure activity relationship (SAR) around the A- and C-rings of the core scaffold. A strategy was developed wherein R7 and R8 A-ring substituents were incorporated late in the synthetic sequence by utilizing palladium-catalyzed cross-coupling reactions on appropriate bromo precursors. Analogues display IC50 values as low as 210 nM and reveal A-ring substituents that enhance selectivity toward either kinase. In cell assays, selected analogues display enhanced phosphorylation of p38 or TBK1 and elicited IL-6 secretion in 3T3-L1 adipocytes better than amlexanox. An analogue bearing a R7 cyclohexyl modification demonstrated robust IL-6 production in 3T3-L1 cells as well as a phosphorylation marker of efficacy and was tested in obese mice where it promoted serum IL-6 response, weight loss, and insulin sensitizing effects comparable to amlexanox. These studies provide impetus to expand the SAR around the amlexanox core toward uncovering analogues with development potential.
中文翻译:

取代的 2-氨基-5-氧代-5H-色并[2,3-b]吡啶-3-羧酸衍生物的设计、合成和生物活性作为炎症激酶 TBK1 和 IKKε 的抑制剂用于治疗肥胖
非经典 IκB 激酶 TANK 结合激酶 1 (TBK1) 和核因子 kappa-B 激酶 ε 抑制剂 (IKKε) 在胰岛素依赖性途径中发挥着关键作用,可促进能量储存并阻止肥胖期间的适应性能量消耗。利用对接计算和 TBK1 与 amlexanox(这些激酶的抑制剂,具有中等效力)结合的 X 射线结构,合成了一系列类似物,以开发核心 A 环和 C 环周围的结构活性关系 (SAR)脚手架。开发了一种策略,其中通过在适当的溴前体上利用钯催化的交叉偶联反应,将R 7和R 8 A环取代基并入合成顺序的后期。类似物的 IC 50值低至 210 nM,并揭示了 A 环取代基,可增强对任一激酶的选择性。在细胞测定中,选定的类似物显示出增强的 p38 或 TBK1 磷酸化,并比 amlexanox 更好地引发 3T3-L1 脂肪细胞中的 IL-6 分泌。带有 R 7环己基修饰的类似物在 3T3-L1 细胞中表现出强大的 IL-6 产生以及磷酸化功效标记物,并在肥胖小鼠中进行了测试,其促进血清 IL-6 反应、体重减轻和胰岛素增敏效果相当氨来呫诺。这些研究为扩大氨来呫诺核心的 SAR 提供动力,以发现具有开发潜力的类似物。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号