当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Self-assembly of penta-selenopeptides into amyloid fibrils†
Chemical Communications ( IF 4.3 ) Pub Date : 2018-09-14 00:00:00 , DOI: 10.1039/c8cc06528d Ram P. Gokula 1, 2, 3, 4 , Jaladhar Mahato 1, 2, 3, 4 , Harkesh B. Singh 1, 2, 3, 4 , Arindam Chowdhury 1, 2, 3, 4
Chemical Communications ( IF 4.3 ) Pub Date : 2018-09-14 00:00:00 , DOI: 10.1039/c8cc06528d Ram P. Gokula 1, 2, 3, 4 , Jaladhar Mahato 1, 2, 3, 4 , Harkesh B. Singh 1, 2, 3, 4 , Arindam Chowdhury 1, 2, 3, 4
Affiliation
|
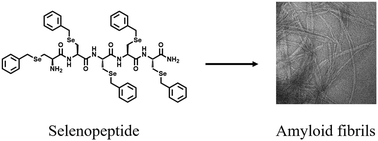
Here, we report the synthesis of a penta-selenopeptide consisting of five benzyl protected selenocysteine residues. This selenopeptide was well characterized by both one- and two-dimensional (D) NMR spectroscopies. We find that the solution conformation is enriched with β-sheet structures, which have a propensity to self-assemble and form amyloid fibrils.
中文翻译:

五硒肽自组装成淀粉样原纤维†
在这里,我们报告由五个苄基保护的硒代半胱氨酸残基组成的五硒肽的合成。该硒肽通过一维和二维(D)NMR光谱学得到了很好的表征。我们发现溶液构象富含β-折叠结构,其具有自组装和形成淀粉样原纤维的倾向。
更新日期:2018-09-14
中文翻译:

五硒肽自组装成淀粉样原纤维†
在这里,我们报告由五个苄基保护的硒代半胱氨酸残基组成的五硒肽的合成。该硒肽通过一维和二维(D)NMR光谱学得到了很好的表征。我们发现溶液构象富含β-折叠结构,其具有自组装和形成淀粉样原纤维的倾向。













































 京公网安备 11010802027423号
京公网安备 11010802027423号