Tetrahedron Letters ( IF 1.5 ) Pub Date : 2018-06-05 , DOI: 10.1016/j.tetlet.2018.06.008 Li Zhang , Mingchao Tan , Lihong Zhou , Qingle Zeng
|
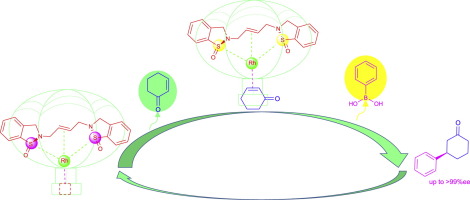
A C2-symmetric, chiral bis-cyclosulfinamide-olefin ligand composed of two 1-oxo-2,3-dihydro-1,2-benzisothiazole moieties with rigid skeletons and a conformationally flexible butenylene chain is disclosed for the first time. HRMS and 1H NMR analyses verify that the in situ-generated complex of the ligand and [Rh(C2H4)2Cl]2 possesses a rhodium (I) center coordinated to the tridentate ligand via two sulfinyl moieties and a CdbndC bond. The chiral ligand provided extremely high enantioselectivity (up to >99%ee) in the Rh-catalyzed asymmetric 1,4-additions of arylboronic acids to cyclohexenone and cyclopentenone. The tridentate ligand gave much higher enantioselectivity than the analogous chiral bidentate ligands.
中文翻译:

Rh催化的不对称1,4-加成反应中的新型C 2对称的手性双环亚磺酰胺-烯烃三齿配体
首次公开了由两个具有刚性骨架和构象柔性的丁烯链的1-氧代-2,3-二氢-1,2-苯并噻唑结构部分组成的AC 2对称的手性双环亚磺酰胺-烯烃配体。HRMS和1 H NMR分析证实,该配体与[Rh(C 2 H 4)2 Cl] 2原位生成的络合物具有通过两个亚磺酰基部分和一个CdbndC键与三齿配体配位的铑(I)中心。手性配体在Rh催化的芳基硼酸与环己烯酮和环戊烯酮的不对称1,4-加成反应中提供了极高的对映选择性(高达> 99%ee)。与类似的手性二齿配体相比,三齿配体具有更高的对映选择性。













































 京公网安备 11010802027423号
京公网安备 11010802027423号