当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
A sterically shielded, stabilized nitrile imine for rapid bioorthogonal protein labeling in live cells
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2018-03-22 , DOI: 10.1021/jacs.8b00126
Peng An 1 , Tracey M. Lewandowski 1 , Tuğçe G. Erbay 2 , Peng Liu 2 , Qing Lin 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2018-03-22 , DOI: 10.1021/jacs.8b00126
Peng An 1 , Tracey M. Lewandowski 1 , Tuğçe G. Erbay 2 , Peng Liu 2 , Qing Lin 1
Affiliation
|
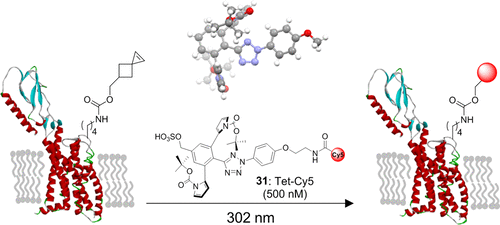
In pursuit of fast bioorthogonal reactions, reactive moieties have been increasingly employed for selective labeling of biomolecules in living systems, posing a challenge in attaining reactivity without sacrificing selectivity. To address this challenge, here we report a bioinspired strategy in which molecular shape controls the selectivity of a transient, highly reactive nitrile imine dipole. By tuning the shape of structural pendants attached to the ortho position of the N-aryl ring of diaryltetrazoles-precursors of nitrile imines, we discovered a sterically shielded nitrile imine that favors the 1,3-dipolar cycloaddition over the competing nucleophilic addition. The photogenerated nitrile imine exhibits an extraordinarily long half-life of 102 s in aqueous medium, owing to its unique molecular shape that hinders the approach of a nucleophile as shown by DFT calculations. The utility of this sterically shielded nitrile imine in rapid (∼1 min) bioorthogonal labeling of glucagon receptor in live mammalian cells was demonstrated.
中文翻译:

空间屏蔽、稳定的腈亚胺,用于活细胞中的快速生物正交蛋白质标记
为了追求快速的生物正交反应,越来越多地采用反应性部分来选择性标记生命系统中的生物分子,这对在不牺牲选择性的情况下获得反应性提出了挑战。为了应对这一挑战,我们在此报告了一种仿生策略,其中分子形状控制瞬态、高反应性腈亚胺偶极子的选择性。通过调整连接在二芳基四唑(腈亚胺前体)的 N-芳基环邻位上的结构悬垂物的形状,我们发现了一种空间屏蔽的腈亚胺,它有利于 1,3-偶极环加成而不是竞争性亲核加成。光生腈亚胺在水性介质中具有 102 秒的超长半衰期,由于其独特的分子形状阻碍了亲核试剂的接近,如 DFT 计算所示。证明了这种空间屏蔽的腈亚胺在活哺乳动物细胞中胰高血糖素受体的快速(~1 分钟)生物正交标记中的效用。
更新日期:2018-03-22
中文翻译:

空间屏蔽、稳定的腈亚胺,用于活细胞中的快速生物正交蛋白质标记
为了追求快速的生物正交反应,越来越多地采用反应性部分来选择性标记生命系统中的生物分子,这对在不牺牲选择性的情况下获得反应性提出了挑战。为了应对这一挑战,我们在此报告了一种仿生策略,其中分子形状控制瞬态、高反应性腈亚胺偶极子的选择性。通过调整连接在二芳基四唑(腈亚胺前体)的 N-芳基环邻位上的结构悬垂物的形状,我们发现了一种空间屏蔽的腈亚胺,它有利于 1,3-偶极环加成而不是竞争性亲核加成。光生腈亚胺在水性介质中具有 102 秒的超长半衰期,由于其独特的分子形状阻碍了亲核试剂的接近,如 DFT 计算所示。证明了这种空间屏蔽的腈亚胺在活哺乳动物细胞中胰高血糖素受体的快速(~1 分钟)生物正交标记中的效用。































 京公网安备 11010802027423号
京公网安备 11010802027423号