当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Histidine-Specific Peptide Modification via Visible-Light-Promoted C-H Alkylation
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2019-10-21 , DOI: 10.1021/jacs.9b09127 Xiaoping Chen 1 , Farong Ye 1 , Xiaosheng Luo 1 , Xueyi Liu 1 , Jie Zhao 1 , Siyao Wang 1 , Qingqing Zhou 1 , Gong Chen 2 , Ping Wang 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2019-10-21 , DOI: 10.1021/jacs.9b09127 Xiaoping Chen 1 , Farong Ye 1 , Xiaosheng Luo 1 , Xueyi Liu 1 , Jie Zhao 1 , Siyao Wang 1 , Qingqing Zhou 1 , Gong Chen 2 , Ping Wang 1
Affiliation
|
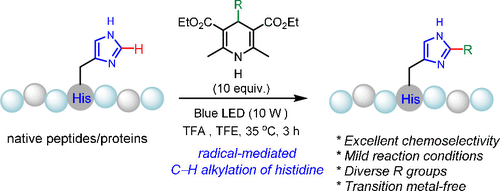
Histidine (His) carries a unique heteroaromatic imidazole side chain and plays irreplaceable functional roles in peptides and proteins. Existing strategies for site-selective histidine modification predominantly rely on the N-substitution reactions of the moderately nucleophilic imidazole group, which inherently suffers from the interferences from lysine and cysteine residues. Chemoselective modification of histidine remain one of the most difficult challenges in peptide chemistry. Herein, we report peptide modification via radical-mediated chemoselective C-H alkylation of histidine using C4-alkyl-1,4-dihydropyridine (DHP) reagents under visible light promoted conditions. The method exploits the electrophilic reactivity of the imidazole ring via a Minisci-type reaction pathway. This method exhibits an exceptionally broad scope for both peptides and DHP alkylation reagents. Its utility has been demonstrated in a series of important peptide drugs, complex natural products, and a small protein. Distinct from N-substitution reactions, the unsubstituted nitrogen groups of the modified imidazole ring are conserved in the C-H alkylated products.
中文翻译:

通过可见光促进的 CH 烷基化修饰组氨酸特异性肽
组氨酸(His)带有独特的杂芳族咪唑侧链,在多肽和蛋白质中发挥着不可替代的功能作用。现有的位点选择性组氨酸修饰策略主要依赖于适度亲核的咪唑基团的 N 取代反应,其固有地受到赖氨酸和半胱氨酸残基的干扰。组氨酸的化学选择性修饰仍然是肽化学中最困难的挑战之一。在此,我们报告了在可见光促进条件下使用 C4-烷基-1,4-二氢吡啶 (DHP) 试剂通过自由基介导的化学选择性 CH 烷基化组氨酸进行肽修饰。该方法通过 Minisci 型反应途径利用咪唑环的亲电反应性。该方法对肽和 DHP 烷基化试剂表现出异常广泛的范围。其效用已在一系列重要的肽药物、复杂的天然产物和小蛋白质中得到证明。与 N 取代反应不同,改性咪唑环的未取代氮基团在 CH 烷基化产物中是保守的。
更新日期:2019-10-21
中文翻译:

通过可见光促进的 CH 烷基化修饰组氨酸特异性肽
组氨酸(His)带有独特的杂芳族咪唑侧链,在多肽和蛋白质中发挥着不可替代的功能作用。现有的位点选择性组氨酸修饰策略主要依赖于适度亲核的咪唑基团的 N 取代反应,其固有地受到赖氨酸和半胱氨酸残基的干扰。组氨酸的化学选择性修饰仍然是肽化学中最困难的挑战之一。在此,我们报告了在可见光促进条件下使用 C4-烷基-1,4-二氢吡啶 (DHP) 试剂通过自由基介导的化学选择性 CH 烷基化组氨酸进行肽修饰。该方法通过 Minisci 型反应途径利用咪唑环的亲电反应性。该方法对肽和 DHP 烷基化试剂表现出异常广泛的范围。其效用已在一系列重要的肽药物、复杂的天然产物和小蛋白质中得到证明。与 N 取代反应不同,改性咪唑环的未取代氮基团在 CH 烷基化产物中是保守的。





















































 京公网安备 11010802027423号
京公网安备 11010802027423号