Nature Energy ( IF 49.7 ) Pub Date : 2019-09-02 , DOI: 10.1038/s41560-019-0451-x Chuan Xia , Peng Zhu , Qiu Jiang , Ying Pan , Wentao Liang , Eli Stavitski , Husam N. Alshareef , Haotian Wang
|
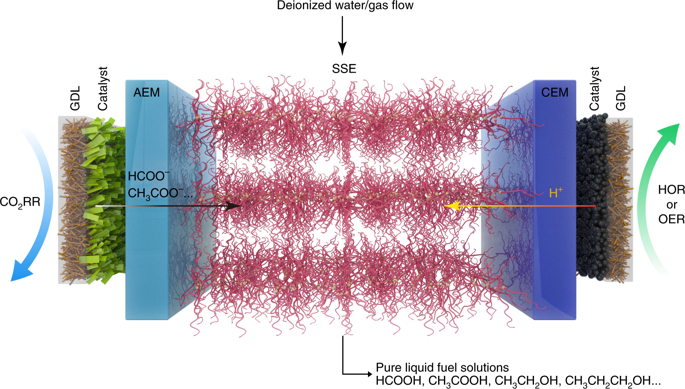
Electrocatalytic CO2 reduction is often carried out in a solution electrolyte such as KHCO3(aq), which allows for ion conduction between electrodes. Therefore, liquid products that form are in a mixture with the dissolved salts, requiring energy-intensive downstream separation. Here, we report continuous electrocatalytic conversion of CO2 to pure liquid fuel solutions in cells that utilize solid electrolytes, where electrochemically generated cations (such as H+) and anions (such as HCOO−) are combined to form pure product solutions without mixing with other ions. Using a HCOOH-selective (Faradaic efficiencies > 90%) and easily scaled Bi catalyst at the cathode, we demonstrate production of pure HCOOH solutions with concentrations up to 12 M. We also show 100 h continuous and stable generation of 0.1 M HCOOH with negligible degradation in selectivity and activity. Production of other electrolyte-free C2+ liquid oxygenate solutions, including acetic acid, ethanol and n-propanol, are also demonstrated using a Cu catalyst. Finally, we show that our CO2 reduction cell with solid electrolytes can be modified to suit other, more complex practical applications.
中文翻译:

使用固体电解质装置通过电催化还原CO 2连续生产纯液体燃料溶液
电催化CO 2还原通常在诸如KHCO 3(aq)的溶液电解质中进行,这允许电极之间的离子传导。因此,形成的液体产物与溶解的盐混合在一起,需要进行能量密集的下游分离。这里,我们报告CO的连续电催化转化2在利用固体电解质电池,以纯液体燃料的解决方案,其中电化学产生阳离子(例如H +)和阴离子(如HCOO -)合并后可形成纯净的产品溶液,而无需与其他离子混合。在阴极上使用选择性HCOOH(法拉第效率> 90%)和易于缩放的Bi催化剂,我们证明了浓度高达12 M的纯HCOOH溶液的生产。我们还显示了100 h连续,稳定地产生0.1 M HCOOH且可忽略不计选择性和活性下降。还证明了使用Cu催化剂生产其他无电解质的C 2+液态含氧溶液,包括乙酸,乙醇和正丙醇。最后,我们表明可以对带有固体电解质的CO 2还原电池进行改造,以适应其他更复杂的实际应用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号