当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Ruthenium-catalyzed ortho-selective CAr–H amination of heteroaryl arenes with di-tert-butyldiaziridinone†
Chemical Communications ( IF 4.3 ) Pub Date : 2019-04-11 00:00:00 , DOI: 10.1039/c9cc02499a Xue-Ya Gou 1, 2, 3, 4 , Yuke Li 5, 6, 7, 8 , Xin-Gang Wang 1, 2, 3, 4 , Hong-Chao Liu 1, 2, 3, 4 , Bo-Sheng Zhang 1, 2, 3, 4 , Jia-Hui Zhao 1, 2, 3, 4 , Zhao-Zhao Zhou 1, 2, 3, 4 , Yong-Min Liang 1, 2, 3, 4
Chemical Communications ( IF 4.3 ) Pub Date : 2019-04-11 00:00:00 , DOI: 10.1039/c9cc02499a Xue-Ya Gou 1, 2, 3, 4 , Yuke Li 5, 6, 7, 8 , Xin-Gang Wang 1, 2, 3, 4 , Hong-Chao Liu 1, 2, 3, 4 , Bo-Sheng Zhang 1, 2, 3, 4 , Jia-Hui Zhao 1, 2, 3, 4 , Zhao-Zhao Zhou 1, 2, 3, 4 , Yong-Min Liang 1, 2, 3, 4
Affiliation
|
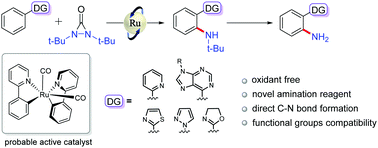
Application of an oxidative amination reagent (di-tert-butyldiaziridinone) to the Ru3(CO)12-catalyzed ortho-selective CAr–H amination reaction is described. This strategy shows good functional group compatibility with various phenyl-substituted N-heterocycles, including biologically active substrates, thus providing synthetic potential for this methodology. Mechanistic studies showed that the reaction process involves an octahedral ruthenium species, and the carbon monoxide ligand plays a crucial role in the C–H activation.
中文翻译:

钌催化的杂芳基芳烃与二叔丁基二氮杂吡啶酮的邻位选择性C Ar –H胺化†
描述了在Ru 3(CO)12催化的邻位选择性C Ar –H胺化反应中使用氧化胺化试剂(二叔丁基二氮并吡啶酮)。该策略显示了与各种苯基取代的N-杂环(包括生物活性底物)的良好官能团相容性,因此为该方法提供了合成潜力。机理研究表明,反应过程涉及八面体钌物种,一氧化碳配体在C–H活化中起关键作用。
更新日期:2019-04-11
中文翻译:

钌催化的杂芳基芳烃与二叔丁基二氮杂吡啶酮的邻位选择性C Ar –H胺化†
描述了在Ru 3(CO)12催化的邻位选择性C Ar –H胺化反应中使用氧化胺化试剂(二叔丁基二氮并吡啶酮)。该策略显示了与各种苯基取代的N-杂环(包括生物活性底物)的良好官能团相容性,因此为该方法提供了合成潜力。机理研究表明,反应过程涉及八面体钌物种,一氧化碳配体在C–H活化中起关键作用。













































 京公网安备 11010802027423号
京公网安备 11010802027423号