European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2019-03-24 , DOI: 10.1016/j.ejmech.2019.03.047 Guo-qing Lu , Xin-yang Li , Kamara Mohamed O , Depu Wang , Fan-hao Meng
|
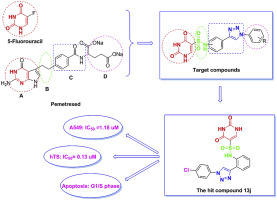
Research on thymidylate synthase inhibitors has been a hot spot for anticancer drug development. Here, based on the structures and pharmacological properties of two types of TS inhibitors, through a molecular assembly principle of drugs design, we designed and synthesized a series of 30 novel uracil derivatives as TS inhibitors. The antiproliferative ability of these compounds was evaluated against four cancer cell lines (A549, OVCAR-3, SGC-7901, and HepG2) by the MTT assay. Most of them showed excellent activities against all the tested cell lines. Furthermore, hTS assay results showed that these compounds have the unique ability to inhibit hTS activity in vitro. Notably, compound 13j exhibited the most potent activity against A549 cells (IC50 = 1.18 μM) and extremely prominent enzyme inhibition (IC50 = 0.13 μM), which was superior to the pemetrexed (PTX, IC50 = 3.29 μM and IC50 = 2.04 μM). Flow cytometric analysis showed the compound 13j could inhibit A549 cells proliferation by arresting the cell cycle in the G1/S phase, then induced the cell apoptosis. Further western blot analysis showed that compound 13j could down-regulate the cycle checkpoint proteins cyclin D1 and cyclin E to inhibit the cell cycle progression, and then induce intrinsic apoptosis by activating caspase-3, and reducing the ratio of bcl-2/bax. All of these results demonstrated that this new structure has potential drug-making properties and provides new ideas for drug development.
中文翻译:

设计,合成和生物学评估带有1、2、3-三唑部分的新型尿嘧啶衍生物作为胸苷酸合酶(TS)抑制剂和潜在的抗肿瘤药物
胸苷酸合酶抑制剂的研究一直是抗癌药物开发的热点。在此,根据两种TS抑制剂的结构和药理特性,通过药物设计的分子组装原理,我们设计合成了30种新颖的尿嘧啶衍生物作为TS抑制剂。通过MTT分析评估了这些化合物对四种癌细胞系(A549,OVCAR-3,SGC-7901和HepG2)的抗增殖能力。它们中的大多数对所有测试的细胞系均表现出优异的活性。此外,hTS分析结果表明,这些化合物具有独特的体外抑制hTS活性的能力。值得注意的是,化合物13j对A549细胞表现出最强的活性(IC 50 = 1.18μM)和极显着的酶抑制(IC 50 = 0.13μM),优于培美曲塞(PTX,IC 50 = 3.29μM和IC 50 = 2.04μM)。流式细胞仪分析表明,化合物13j可以通过将细胞周期阻滞在G1 / S期来抑制A549细胞的增殖,进而诱导细胞凋亡。进一步的蛋白质印迹分析表明化合物13j可能下调周期检查点蛋白cyclin D1和cyclin E以抑制细胞周期进程,然后通过激活caspase-3并降低bcl-2 / bax的比例来诱导内在凋亡。所有这些结果表明,这种新结构具有潜在的药物制造特性,并为药物开发提供了新思路。































 京公网安备 11010802027423号
京公网安备 11010802027423号