理由:越来越多的证据表明,细胞内活性氧(ROS)的积累是骨质疏松症发展的关键因素,它会触发破骨细胞的形成和功能。伪神经素A(Pseurotin A,Pse)是从烟曲霉分离的具有抗氧化特性的次生代谢产物,最近被证明具有广泛的潜在治疗应用。但是,其对骨质疏松症的作用仍然未知。这项研究旨在探讨Pse通过抑制ROS水平是否能够抑制卵巢切除(OVX)小鼠中的破骨细胞生成并预防由雌激素缺乏引起的骨质流失。
方法:通过抗酒石酸酸性磷酸酶(TRAcP)染色和羟磷灰石吸收试验,研究了Pse对核因子-κB(NF-κB)配体(RANKL)诱导的破骨细胞形成和骨吸收功能的受体激活剂的影响。2',7'-二氯二氢荧光素二乙酸酯(H 2 DCFDA)用于检测体外细胞内ROS的产生。Western blot分析用于鉴定与ROS产生和清除相关的蛋白,以及ROS介导的信号级联反应,包括有丝分裂原激活的蛋白激酶(MAPK),NF-κB途径和活化T细胞1(NFATc1)信号的核因子。通过qPCR评估破骨细胞特异性基因的表达。在体内使用给予Pse或媒介物6周的OVX小鼠模型确定Pse的潜力。体内ROS的产生是通过在杀死前24h将二氢乙啶(DHE)静脉注射到OVX小鼠中来评估的。处死后,使用微型CT和组织形态计量学分析骨样品,以确定离体的骨体积,破骨细胞活性和ROS水平。
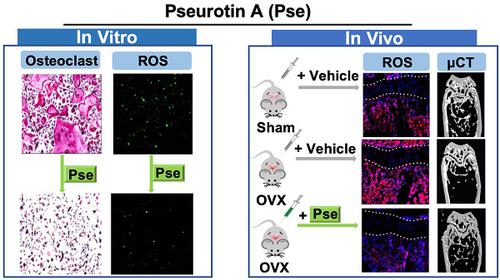
结果: Pse被证明在体外抑制破骨细胞生成和骨吸收功能,以及破骨细胞特异性基因(包括Acp5(编码TRAcP),Ctsk(编码组织蛋白酶K)和Mmp9)的下调。(编码基质金属蛋白酶9)。从机制上讲,Pse通过抑制RANKL诱导的ROS产生并增强ROS清除酶来抑制细胞内ROS水平,随后抑制MAPK途径(ERK,P38和JNK)和NF-κB途径,从而抑制NFATc1信号传导。显微CT和组织学数据表明,OVX手术导致了严重的骨质流失,并显着增加了骨表面破骨细胞的数量以及骨髓微环境中ROS的水平;而补充Pse能够有效预防这些OVX引起的变化。
结论: Pse首次被证明是通过抑制ROS水平来治疗破骨细胞相关性骨疾病(如骨质疏松症)的新型替代疗法。
 "点击查看英文标题和摘要"
"点击查看英文标题和摘要"
Pseurotin A Inhibits Osteoclastogenesis and Prevents Ovariectomized-Induced Bone Loss by Suppressing Reactive Oxygen Species
Rationale: Growing evidence indicates that intracellular reactive oxygen species (ROS) accumulation is a critical factor in the development of osteoporosis by triggering osteoclast formation and function. Pseurotin A (Pse) is a secondary metabolite isolated from Aspergillus fumigatus with antioxidant properties, recently shown to exhibit a wide range of potential therapeutic applications. However, its effects on osteoporosis remain unknown. This study aimed to explore whether Pse, by suppressing ROS level, is able to inhibit osteoclastogenesis and prevent the bone loss induced by estrogen-deficiency in ovariectomized (OVX) mice.
Methods: The effects of Pse on receptor activator of nuclear factor-κB (NF-κB) ligand (RANKL)-induced osteoclastogenesis and bone resorptive function were examined by tartrate resistant acid phosphatase (TRAcP) staining and hydroxyapatite resorption assay. 2',7'-dichlorodihydrofluorescein diacetate (H2DCFDA) was used to detect intracellular ROS production in vitro. Western blot assay was used to identify proteins associated with ROS generation and scavenging as well as ROS-mediated signaling cascades including mitogen-activated protein kinases (MAPKs), NF-κB pathways, and nuclear factor of activated T cells 1 (NFATc1) signaling. The expression of osteoclast-specific genes was assessed by qPCR. The in vivo potential of Pse was determined using an OVX mouse model administered with Pse or vehicle for 6 weeks. In vivo ROS production was assessed by intravenous injection of dihydroethidium (DHE) into OVX mice 24h prior to killing. After sacrifice, the bone samples were analyzed using micro-CT and histomorphometry to determine bone volume, osteoclast activity, and ROS level ex vivo.
Results: Pse was demonstrated to inhibit osteoclastogenesis and bone resorptive function in vitro, as well as the downregulation of osteoclast-specific genes including Acp5 (encoding TRAcP), Ctsk (encoding cathepsin K), and Mmp9 (encoding matrix metalloproteinase 9). Mechanistically, Pse suppressed intracellular ROS level by inhibiting RANKL-induced ROS production and enhancing ROS scavenging enzymes, subsequently suppressing MAPK pathway (ERK, P38, and JNK) and NF-κB pathways, leading to the inhibition of NFATc1 signaling. Micro-CT and histological data indicated that OVX procedure resulted in a significant bone loss, with dramatically increased the number of osteoclasts on the bone surface as well as increased ROS level in the bone marrow microenvironment; whereas Pse supplementation was capable of effectively preventing these OVX-induced changes.
Conclusion: Pse was demonstrated for the first time as a novel alternative therapy for osteoclast-related bone diseases such as osteoporosis through suppressing ROS level.








































 京公网安备 11010802027423号
京公网安备 11010802027423号