当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Amide Enolate Additions to Acylsilanes: In Situ Generation of Unusual and Stereoselective Homoenolate Equivalents
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2009-07-01 , DOI: 10.1021/ja808811u
Robert B. Lettan 1 , Chris V. Galliford 1 , Chase C. Woodward 1 , Karl A. Scheidt 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2009-07-01 , DOI: 10.1021/ja808811u
Robert B. Lettan 1 , Chris V. Galliford 1 , Chase C. Woodward 1 , Karl A. Scheidt 1
Affiliation
|
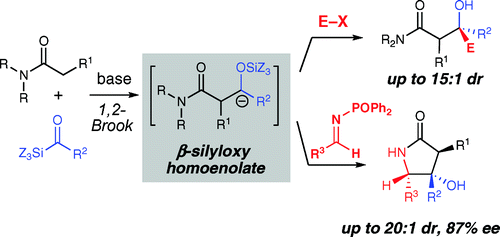
The synthesis of beta-hydroxy carbonyl compounds is an important goal due to their prevalence in bioactive molecules. A novel approach to construct these structural motifs involves the multicomponent reaction of acylsilanes, amides, and electrophiles. The addition of amide enolates to acylsilanes generates beta-silyloxy homoenolate reactivity by undergoing a 1,2-Brook rearrangement. These unique nucleophiles formed in situ can then undergo addition to alkyl halides, aldehydes, ketones, and imines. The gamma-amino-beta-hydroxy amide products derived from the addition of these homoenolates to N-diphenylphosphinyl imines are generated with excellent diastereoselectivity (> or = 20:1) and can be efficiently converted to highly valuable gamma-lactams. Finally, the use of optically active amide enolates delivers beta-hydroxy amide products with high levels of diastereoselectivity (> or = 10:1).
中文翻译:

酰胺烯醇化物添加到酰基硅烷:原位生成异常和立体选择性均烯醇化物等价物
β-羟基羰基化合物的合成是一个重要的目标,因为它们普遍存在于生物活性分子中。一种构建这些结构基序的新方法涉及酰基硅烷、酰胺和亲电试剂的多组分反应。通过进行 1,2-布鲁克重排,将酰胺烯醇化物添加到酰基硅烷中会产生 β-甲硅烷氧基均烯醇化物反应性。这些在原位形成的独特亲核试剂随后可以与卤代烷、醛、酮和亚胺进行加成。通过将这些同烯醇化物添加到 N-二苯基膦亚胺中而产生的 γ-氨基-β-羟基酰胺产物具有出色的非对映选择性(> 或 = 20:1),并且可以有效地转化为高价值的 γ-内酰胺。最后,
更新日期:2009-07-01
中文翻译:

酰胺烯醇化物添加到酰基硅烷:原位生成异常和立体选择性均烯醇化物等价物
β-羟基羰基化合物的合成是一个重要的目标,因为它们普遍存在于生物活性分子中。一种构建这些结构基序的新方法涉及酰基硅烷、酰胺和亲电试剂的多组分反应。通过进行 1,2-布鲁克重排,将酰胺烯醇化物添加到酰基硅烷中会产生 β-甲硅烷氧基均烯醇化物反应性。这些在原位形成的独特亲核试剂随后可以与卤代烷、醛、酮和亚胺进行加成。通过将这些同烯醇化物添加到 N-二苯基膦亚胺中而产生的 γ-氨基-β-羟基酰胺产物具有出色的非对映选择性(> 或 = 20:1),并且可以有效地转化为高价值的 γ-内酰胺。最后,

































 京公网安备 11010802027423号
京公网安备 11010802027423号