当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Deiodination of Thyroid Hormones by Iodothyronine Deiodinase Mimics: Does an Increase in the Reactivity Alter the Regioselectivity?
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2011-07-06 , DOI: 10.1021/ja201657s Debasish Manna 1 , Govindasamy Mugesh 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2011-07-06 , DOI: 10.1021/ja201657s Debasish Manna 1 , Govindasamy Mugesh 1
Affiliation
|
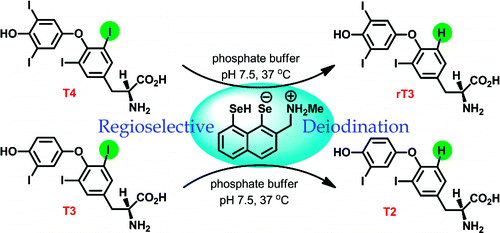
Organoselenium compounds as functional mimics of iodothyronine deiodinase are described. The naphthyl-based compounds having two selenol groups are remarkably efficient in the inner-ring deiodination of thyroxine. The introduction of a basic amino group in close proximity to one of the selenol moieties enhances the deiodination. This study suggests that an increase in the nucleophilic reactivity of the conserved Cys residue at the active site of deiodinases is very important for effective deiodination.
中文翻译:

碘甲状腺原氨酸脱碘酶模拟物对甲状腺激素的脱碘:反应性的增加是否会改变区域选择性?
描述了作为碘甲状腺原氨酸脱碘酶功能模拟物的有机硒化合物。具有两个硒醇基团的萘基化合物在甲状腺素的内环脱碘中非常有效。在硒醇部分之一附近引入碱性氨基可增强脱碘作用。该研究表明,脱碘酶活性位点上保守的 Cys 残基亲核反应性的增加对于有效脱碘非常重要。
更新日期:2011-07-06
中文翻译:

碘甲状腺原氨酸脱碘酶模拟物对甲状腺激素的脱碘:反应性的增加是否会改变区域选择性?
描述了作为碘甲状腺原氨酸脱碘酶功能模拟物的有机硒化合物。具有两个硒醇基团的萘基化合物在甲状腺素的内环脱碘中非常有效。在硒醇部分之一附近引入碱性氨基可增强脱碘作用。该研究表明,脱碘酶活性位点上保守的 Cys 残基亲核反应性的增加对于有效脱碘非常重要。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号