当前位置:
X-MOL 学术
›
J. Med. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Group-Based Optimization of Potent and Cell-Active Inhibitors of the von Hippel–Lindau (VHL) E3 Ubiquitin Ligase: Structure–Activity Relationships Leading to the Chemical Probe (2S,4R)-1-((S)-2-(1-Cyanocyclopropanecarboxamido)-3,3-dimethylbutanoyl)-4-hydroxy-N-(4-(4-methylthiazol-5-yl)benzyl)pyrrolidine-2-carboxamide (VH298)
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2017-09-18 00:00:00 , DOI: 10.1021/acs.jmedchem.7b00675 Pedro Soares 1 , Morgan S. Gadd 1 , Julianty Frost 1, 2 , Carles Galdeano 1 , Lucy Ellis 1 , Ola Epemolu 1 , Sonia Rocha 2 , Kevin D. Read 1 , Alessio Ciulli 1
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2017-09-18 00:00:00 , DOI: 10.1021/acs.jmedchem.7b00675 Pedro Soares 1 , Morgan S. Gadd 1 , Julianty Frost 1, 2 , Carles Galdeano 1 , Lucy Ellis 1 , Ola Epemolu 1 , Sonia Rocha 2 , Kevin D. Read 1 , Alessio Ciulli 1
Affiliation
|
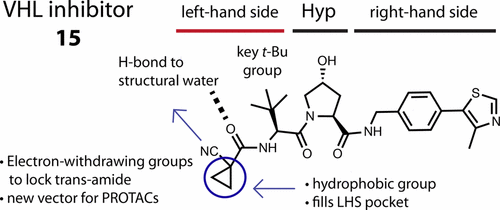
The von Hippel–Lindau tumor suppressor protein is the substrate binding subunit of the VHL E3 ubiquitin ligase, which targets hydroxylated α subunit of hypoxia inducible factors (HIFs) for ubiquitination and subsequent proteasomal degradation. VHL is a potential target for treating anemia and ischemic diseases, motivating the development of inhibitors of the VHL:HIF-α protein–protein interaction. Additionally, bifunctional proteolysis targeting chimeras (PROTACs) containing a VHL ligand can hijack the E3 ligase activity to induce degradation of target proteins. We report the structure-guided design and group-based optimization of a series of VHL inhibitors with low nanomolar potencies and improved cellular permeability. Structure–activity relationships led to the discovery of potent inhibitors 10 and chemical probe VH298, with dissociation constants <100 nM, which induced marked HIF-1α intracellular stabilization. Our study provides new chemical tools to probe the VHL-HIF pathways and new VHL ligands for next-generation PROTACs.
中文翻译:

基于组的von Hippel-Lindau(VHL)E3泛素连接酶有效和细胞活性抑制剂的优化:结构-活性关系导致化学探针(2 S,4 R)-1-((S)-2- (1-氰基环丙烷甲酰胺基)-3,3-二甲基丁酰基)-4-羟基-N-(4-(4-甲基噻唑-5-基)苄基)吡咯烷-2-羧酰胺(VH298)
von Hippel–Lindau肿瘤抑制蛋白是VHL E3泛素连接酶的底物结合亚基,其靶向缺氧诱导因子(HIFs)的羟基化α亚基进行泛素化和随后的蛋白酶体降解。VHL是治疗贫血和缺血性疾病的潜在靶标,从而促进了VHL:HIF-α蛋白与蛋白相互作用抑制剂的发展。此外,包含VHL配体的靶向双功能蛋白水解嵌合体(PROTAC)可以劫持E3连接酶活性,从而诱导目标蛋白降解。我们报告了一系列具有低纳摩尔浓度和改善的细胞渗透性的VHL抑制剂的结构指导设计和基于基团的优化。结构-活性关系导致发现有效的抑制剂10化学探针VH298,解离常数<100 nM,可诱导明显的HIF-1α细胞内稳定。我们的研究提供了新的化学工具,可探测下一代PROTAC的VHL-HIF途径和新的VHL配体。
更新日期:2017-09-18
中文翻译:

基于组的von Hippel-Lindau(VHL)E3泛素连接酶有效和细胞活性抑制剂的优化:结构-活性关系导致化学探针(2 S,4 R)-1-((S)-2- (1-氰基环丙烷甲酰胺基)-3,3-二甲基丁酰基)-4-羟基-N-(4-(4-甲基噻唑-5-基)苄基)吡咯烷-2-羧酰胺(VH298)
von Hippel–Lindau肿瘤抑制蛋白是VHL E3泛素连接酶的底物结合亚基,其靶向缺氧诱导因子(HIFs)的羟基化α亚基进行泛素化和随后的蛋白酶体降解。VHL是治疗贫血和缺血性疾病的潜在靶标,从而促进了VHL:HIF-α蛋白与蛋白相互作用抑制剂的发展。此外,包含VHL配体的靶向双功能蛋白水解嵌合体(PROTAC)可以劫持E3连接酶活性,从而诱导目标蛋白降解。我们报告了一系列具有低纳摩尔浓度和改善的细胞渗透性的VHL抑制剂的结构指导设计和基于基团的优化。结构-活性关系导致发现有效的抑制剂10化学探针VH298,解离常数<100 nM,可诱导明显的HIF-1α细胞内稳定。我们的研究提供了新的化学工具,可探测下一代PROTAC的VHL-HIF途径和新的VHL配体。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号