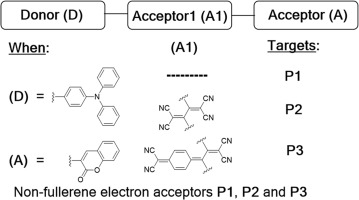
两个简单的半导体施主-受主-受主(d-A 1 -A)模块化的,小分子的,非富勒烯的电子受体,2-(4-(二苯基氨基)苯基)-3-(4 - ((2-氧代-2- H-铬-3-基)乙炔基)苯基)-1,3-二烯-1,1,4,4-四甲腈(P2)和2-(4-(3,3-二氰基-1-(4- (二苯氨基)苯基)-2-(4-((2-氧代2- H-铬-3-基)乙炔基)苯基)亚芳基)环己-2,5-二烯-1-基)丙二腈(P3)。设计,合成和表征了其在可溶液处理的体异质结太阳能电池中的应用。P2和P3的光电和光伏特性直接与结构类似物3-(((4-((4-(二苯氨基)苯基)乙炔基)苯基)乙炔基)-2 H-铬-2--2-酮(P1)的结构类似物进行比较D–A格式。所有这些新材料都包含一个富电子的三苯胺(TPA)供体核(D)和一个贫电子的chromen-2-one末端核(A)。在简单的D–A系统中,TPA和苯醌是最终功能,而在D–A 1 –A系统中,通过保持D / A将四氰基乙烯(TCNE)和四氰基喹二甲烷(TCNQ)衍生的功能合并为A 1单元。单位常数。包含A 1主要是为了诱导分子主链内的交叉共轭并因此产生低带隙靶标。物理和光电特性通过紫外-可见(UV-Vis),热重分析,空气中的电子光谱和循环伏安法进行表征。这些新材料显示出更宽的吸收光谱,例如在P3情况下具有全色吸收,出色的溶解度和热稳定性,并且能级与常规和常规使用的供体聚合物聚(3-己基噻吩)(P3HT)相当。用P1,P2和P3制成可溶液处理的体异质结器件作为非富勒烯电子受体。对光伏性能的研究表明,最佳的基于P3HT:P3的器件显示出令人印象深刻的4.21%的增强的功率转换效率,相对于最佳的基于P3HT:P1的器件的效率(2.28%)提高了大约两倍。)。我们的结果清楚地表明,D–A 1 –A型小分子在有机太阳能电池的研究领域中是很有前途的非富勒烯电子受体。
 "点击查看英文标题和摘要"
"点击查看英文标题和摘要"
Donor–acceptor–acceptor-based non-fullerene acceptors comprising terminal chromen-2-one functionality for efficient bulk-heterojunction devices
Two simple semiconducting donor–acceptor–acceptor (D–A1–A) modular, small molecule, non-fullerene electron acceptors, 2-(4-(diphenylamino)phenyl)-3-(4-((2-oxo-2H-chromen-3-yl)ethynyl)phenyl)buta-1,3-diene-1,1,4,4-tetracarbonitrile (P2) and 2-(4-(3,3-dicyano-1-(4-(diphenylamino)phenyl)-2-(4-((2-oxo-2H-chromen-3-yl)ethynyl)phenyl)allylidene)cyclohexa-2,5-dien-1-ylidene)malononitrile (P3), were designed, synthesized and characterized for application in solution-processable bulk-heterojunction solar cells. The optoelectronic and photovoltaic properties of P2 and P3 were directly compared with those of a structural analogue, 3-((4-((4-(diphenylamino)phenyl)ethynyl)phenyl)ethynyl)-2H-chromen-2-one (P1), which was designed based on a D–A format. All of these new materials comprised an electron rich triphenylamine (TPA) donor core (D) and electron deficient chromen-2-one terminal core (A). In the simple D–A system, TPA and chromenone were the terminal functionalities, whereas in the D–A1–A system, tetracyanoethylene (TCNE) and tetracyanoquinodimethane (TCNQ) derived functionalities were incorporated as A1 units by keeping the D/A units constant. The inclusion of A1 was primarily done to induce cross-conjugation within the molecular backbone and hence to generate low band gap targets. The physical and optoelectronic properties were characterized by ultraviolet–visible (UV–Vis), thermogravimetric analysis, photo-electron spectroscopy in air and cyclic voltammetry. These new materials exhibited broadened absorption spectra, for instance panchromatic absorbance in case of P3, excellent solubility and thermal stability, and energy levels matching those of the conventional and routinely used donor polymer poly(3-hexyl thiophene) (P3HT). Solution-processable bulk-heterojunction devices were fabricated with P1, P2 and P3 as non-fullerene electron acceptors. Studies on the photovoltaic properties revealed that the best P3HT: P3-based device showed an impressive enhanced power conversion efficiency of 4.21%, an increase of around two-fold with respect to the efficiency of the best P3HT: P1-based device (2.28%). Our results clearly demonstrate that the D–A1–A type small molecules are promising non-fullerene electron acceptors in the research field of organic solar cells.












































 京公网安备 11010802027423号
京公网安备 11010802027423号