当前位置:
X-MOL 学术
›
Tetrahedron
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
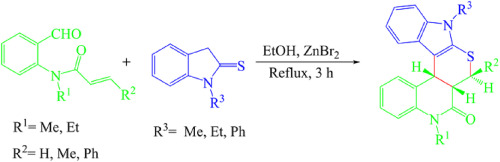
ZnBr2 catalyzed domino Knoevenagel-hetero-Diels–Alder reaction: An efficient route to polycyclic thiopyranoindol annulated [3,4-c]quinolone derivatives
Tetrahedron ( IF 2.1 ) Pub Date : 2017-04-26 06:24:21 Mostafa Kiamehr, Batoul Alipour, Leyla Mohammadkhani, Behzad Jafari, Peter Langer
Tetrahedron ( IF 2.1 ) Pub Date : 2017-04-26 06:24:21 Mostafa Kiamehr, Batoul Alipour, Leyla Mohammadkhani, Behzad Jafari, Peter Langer
|
Various novel polycyclic thiopyranoindol annulated [3,4-c]quinolone derivatives were synthesized via domino Knoevenagel-hetero-Diels–Alder reactions of indoline-2-thions and novel N-acrylated anthranilaldehydes in refluxing ethanol as a solvent in the presence of 20 mol% ZnBr2 as a Lewis acid catalyst. All reactions proceed with high yields with excellent regio- and stereoselectivity.
中文翻译:

ZnBr2催化多米诺Knoevenagel-杂-Diels-Alder反应:一种有效的途径来制备多环硫代吡喃并吲哚环化的[3,4-c]喹诺酮衍生物
在20 mol的乙醇存在下,通过吲哚-2-噻吩的多米诺酮的Knoevenagel-杂Diels-Alder反应和新颖的N-丙烯酸蒽醛在回流的乙醇中作为溶剂合成了各种新的多环硫吡喃吲哚环化的[3,4-c]喹诺酮衍生物。 %ZnBr 2作为路易斯酸催化剂。所有反应均以高产率进行,具有优异的区域选择性和立体选择性。
更新日期:2017-04-26
中文翻译:

ZnBr2催化多米诺Knoevenagel-杂-Diels-Alder反应:一种有效的途径来制备多环硫代吡喃并吲哚环化的[3,4-c]喹诺酮衍生物
在20 mol的乙醇存在下,通过吲哚-2-噻吩的多米诺酮的Knoevenagel-杂Diels-Alder反应和新颖的N-丙烯酸蒽醛在回流的乙醇中作为溶剂合成了各种新的多环硫吡喃吲哚环化的[3,4-c]喹诺酮衍生物。 %ZnBr 2作为路易斯酸催化剂。所有反应均以高产率进行,具有优异的区域选择性和立体选择性。






































 京公网安备 11010802027423号
京公网安备 11010802027423号