当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Selective Synthesis of [12]Cycloparaphenylene
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2009-07-08 , DOI: 10.1002/anie.200902617 Hiroko Takaba , Haruka Omachi , Yosuke Yamamoto , Jean Bouffard , Kenichiro Itami
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2009-07-08 , DOI: 10.1002/anie.200902617 Hiroko Takaba , Haruka Omachi , Yosuke Yamamoto , Jean Bouffard , Kenichiro Itami
|
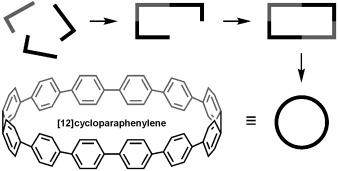
Tighten your belt: [12]Cycloparaphenylene, the sidewall segment of a carbon nanotube (see picture), has been synthesized in a selective manner through stepwise palladium‐catalyzed coupling reactions. The synthesis capitalizes on the ability of the cis‐1,4‐dihydroxycyclohexane‐1,4‐diyl unit to attenuate the build‐up of strain energy during macrocyclization.

中文翻译:

[12]环对亚苯基的选择性合成
系紧安全带:[12]碳纳米管的侧壁部分(见图)环对亚苯基是通过逐步的钯催化的偶联反应以选择性方式合成的。该合成利用顺环1,4-二羟基环己烷-1,4-二基单元在大环化过程中减弱应变能积累的能力。

更新日期:2009-07-08

中文翻译:

[12]环对亚苯基的选择性合成
系紧安全带:[12]碳纳米管的侧壁部分(见图)环对亚苯基是通过逐步的钯催化的偶联反应以选择性方式合成的。该合成利用顺环1,4-二羟基环己烷-1,4-二基单元在大环化过程中减弱应变能积累的能力。












































 京公网安备 11010802027423号
京公网安备 11010802027423号