当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Regiocontrolled Palladium-Catalyzed Arylative Cyclizations of Alkynols
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2014-04-17 , DOI: 10.1021/ja5029028 Daishi Fujino 1 , Hideki Yorimitsu 1, 2 , Atsuhiro Osuka 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2014-04-17 , DOI: 10.1021/ja5029028 Daishi Fujino 1 , Hideki Yorimitsu 1, 2 , Atsuhiro Osuka 1
Affiliation
|
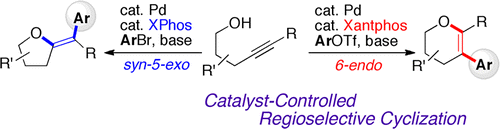
Tuning the reactivity of arylpalladium intermediates enables control of catalytic arylative 5-exo and 6-endo cyclizations of alkynols. The two modes of cyclizations represent a rare example of controllable, regioselective difunctionalization of alkynes. The cyclizations are useful in offering a divergent synthesis of oxygen-containing heterocycles, which is of synthetic use for further derivatization. Formal synthesis of an hNK-1 receptor antagonist also showcases the utility of our arylative cyclization.
中文翻译:

区域控制的钯催化的炔醇芳基环化
调节芳基钯中间体的反应性可以控制炔醇的催化芳基化 5-exo 和 6-endo 环化。这两种环化模式代表了炔烃可控、区域选择性双官能化的罕见例子。环化可用于提供含氧杂环的发散合成,其对于进一步衍生化具有合成用途。hNK-1 受体拮抗剂的正式合成也展示了我们芳基化环化的效用。
更新日期:2014-04-17
中文翻译:

区域控制的钯催化的炔醇芳基环化
调节芳基钯中间体的反应性可以控制炔醇的催化芳基化 5-exo 和 6-endo 环化。这两种环化模式代表了炔烃可控、区域选择性双官能化的罕见例子。环化可用于提供含氧杂环的发散合成,其对于进一步衍生化具有合成用途。hNK-1 受体拮抗剂的正式合成也展示了我们芳基化环化的效用。













































 京公网安备 11010802027423号
京公网安备 11010802027423号