当前位置:
X-MOL 学术
›
ACS Appl. Energy Mater.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Temperature Driven Phase Evolution of FeF3 as Active Material in Lithium-Ion Batteries
ACS Applied Energy Materials ( IF 5.4 ) Pub Date : 2024-08-02 , DOI: 10.1021/acsaem.4c01129 Régis Porhiel 1, 2 , Stéphanie Belin 3 , Fannie Alloin 1, 4 , Cristina Iojoiu 1 , Katia Guérin 2
ACS Applied Energy Materials ( IF 5.4 ) Pub Date : 2024-08-02 , DOI: 10.1021/acsaem.4c01129 Régis Porhiel 1, 2 , Stéphanie Belin 3 , Fannie Alloin 1, 4 , Cristina Iojoiu 1 , Katia Guérin 2
Affiliation
|
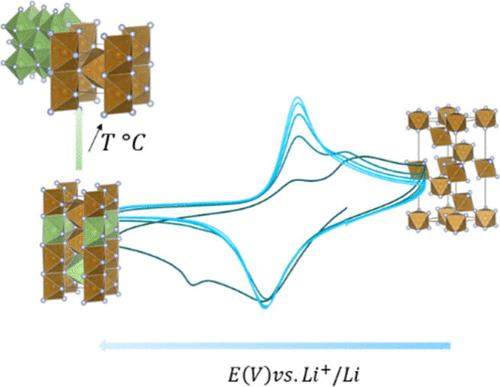
Iron trifluoride as an active material for a lithium-ion battery positive electrode has attracted interest owing to its high theoretical capacity (713 mAh g–1). However, the large volume expansion during cycling and the poor electrical conductivity of the fluoride hinder the reversible performance of FeF3. Moreover, the electrochemical mechanism during reduction and oxidation is not resolved yet, involving complex phase transitions. Recent findings about iron fluorides indicated improved reversibility when the active material is operated with solid polymer electrolytes at temperatures above 60 °C. We herein investigate the electrochemical mechanism of such assembly using operando X-ray absorption spectroscopy along with ex-situ X-ray diffraction for samples with matching liquid electrolytes at several temperatures and selected voltages. We show that the electrochemical mechanism is closely related to the applied temperature, involving distinct reaction pathways between room temperature cells and cells at the temperature required for solid polymer electrolytes. We show that these differences in phases formed at different temperatures are particularly linked to the metastability of the LixFeF3 insertion phase, undergoing a conversion reaction to form FeF2 + LiF at temperatures above 40 °C. This study highlights the need for an in-depth comprehension of the electrochemical mechanism of such materials, notably for understanding the high-temperature performance improvement for FeF3 with solid polymer electrolytes.
中文翻译:

锂离子电池活性材料 FeF3 的温度驱动相演化
三氟化铁作为锂离子电池正极活性材料因其高理论容量(713 mAh g –1 )而引起了人们的兴趣。然而,循环过程中较大的体积膨胀和氟化物较差的导电性阻碍了FeF 3的可逆性能。此外,还原和氧化过程中的电化学机制尚未解决,涉及复杂的相变。关于氟化铁的最新研究结果表明,当活性材料与固体聚合物电解质一起在 60 °C 以上的温度下工作时,可逆性得到改善。我们在本文中使用原位X 射线吸收光谱以及异位 X 射线衍射对在多个温度和选定电压下具有匹配液体电解质的样品研究了这种组装的电化学机制。我们表明,电化学机制与所施加的温度密切相关,涉及室温电池和固体聚合物电解质所需温度下的电池之间不同的反应途径。我们表明,在不同温度下形成的相的这些差异与 Li x FeF 3插入相的亚稳定性特别相关,在高于 40 °C 的温度下发生转化反应形成 FeF 2 + LiF。这项研究强调需要深入理解此类材料的电化学机制,特别是理解固体聚合物电解质对 FeF 3高温性能的改进。
更新日期:2024-08-02
中文翻译:

锂离子电池活性材料 FeF3 的温度驱动相演化
三氟化铁作为锂离子电池正极活性材料因其高理论容量(713 mAh g –1 )而引起了人们的兴趣。然而,循环过程中较大的体积膨胀和氟化物较差的导电性阻碍了FeF 3的可逆性能。此外,还原和氧化过程中的电化学机制尚未解决,涉及复杂的相变。关于氟化铁的最新研究结果表明,当活性材料与固体聚合物电解质一起在 60 °C 以上的温度下工作时,可逆性得到改善。我们在本文中使用原位X 射线吸收光谱以及异位 X 射线衍射对在多个温度和选定电压下具有匹配液体电解质的样品研究了这种组装的电化学机制。我们表明,电化学机制与所施加的温度密切相关,涉及室温电池和固体聚合物电解质所需温度下的电池之间不同的反应途径。我们表明,在不同温度下形成的相的这些差异与 Li x FeF 3插入相的亚稳定性特别相关,在高于 40 °C 的温度下发生转化反应形成 FeF 2 + LiF。这项研究强调需要深入理解此类材料的电化学机制,特别是理解固体聚合物电解质对 FeF 3高温性能的改进。

































 京公网安备 11010802027423号
京公网安备 11010802027423号