Chem Catalysis ( IF 11.5 ) Pub Date : 2024-06-12 , DOI: 10.1016/j.checat.2024.101027 Mengjing Sun , Leiyang Bai , Chengliang Li , Xuefeng Jiang
|
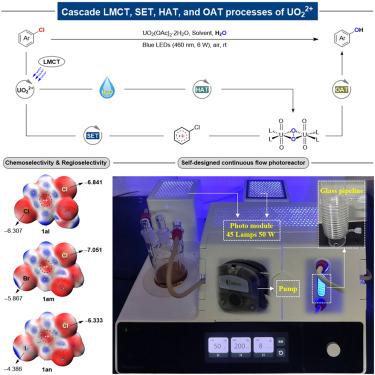
The efficient conversion from aryl chlorides to phenols remains a long-standing challenge with a standard of mild reaction conditions, due to the high energy barrier for C(sp2)−Cl bond activation (Ph−Cl at ∼400 kJ/mol). Herein, we accomplished a facile hydroxylation reaction from unactivated aryl chlorides with water under ambient conditions. The photouranium-catalyzed cascade processes of single-electron transfer, hydrogen atom transfer, and oxygen atom transfer facilitated the synergistic steps of water splitting and cation-radical accelerated nucleophilic aromatic substitution, enabling the late-stage hydroxylation for pharmaceuticals with sensitive functional groups.
中文翻译:

从芳基氯到酚:光铀催化的水分解羟基化
由于 C(sp 2 )−Cl 键激活的高能垒(Ph−Cl at ∼ 400 kJ/摩尔)。在此,我们在环境条件下完成了未活化的芳基氯与水的轻松羟基化反应。光铀催化的单电子转移、氢原子转移和氧原子转移级联过程促进了水分解和阳离子自由基加速亲核芳香取代的协同步骤,从而实现了具有敏感官能团的药物的后期羟基化。






































 京公网安备 11010802027423号
京公网安备 11010802027423号