当前位置:
X-MOL 学术
›
Chin. J. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Chiral Isothiourea-Catalyzed Acylative Dynamic Kinetic Resolution of 3-Hydroxyphthalides for Enantioselective Synthesis of Phthalidyl Esters
Chinese Journal of Chemistry ( IF 5.5 ) Pub Date : 2024-05-27 , DOI: 10.1002/cjoc.202400382
Zeyang Hao 1 , Wei Lin 1 , Zi‐Qi Yuan 1 , Wei Zhang 2 , Xin Li 1
Chinese Journal of Chemistry ( IF 5.5 ) Pub Date : 2024-05-27 , DOI: 10.1002/cjoc.202400382
Zeyang Hao 1 , Wei Lin 1 , Zi‐Qi Yuan 1 , Wei Zhang 2 , Xin Li 1
Affiliation
|
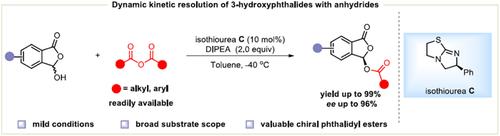
Phthalides serve as core structures pervasive in a wide array of natural products and drug molecules, which display a diverse array of biological activities. We report herein a highly efficient dynamic kinetic resolution of 3-hydroxyphthalides by chiral isothioureas (ITUs) catalyzed asymmetric acylation, facilitating the effective synthesis of a variety of chiral phthalidyl esters with good yields and enantioselectivities. Notably, this reaction features mild reaction conditions, expansive substrate scope as well as good functional group compatibility. In addition, the practicality of this method is underscored by the large-scale synthesis, reduced catalyst loading experiment and the synthesis of the chiral phthalidyl ester prodrug.
中文翻译:

手性异硫脲催化 3-羟基苯酞的酰化动态动力学拆分用于对映选择性合成邻苯二甲酸酯
邻苯二甲酸酯作为核心结构广泛存在于各种天然产物和药物分子中,表现出多种生物活性。我们在此报告了通过手性异硫脲(ITU)催化的不对称酰化对3-羟基苯酞进行高效动态动力学拆分,促进了各种手性苯酞酯的有效合成,具有良好的产率和对映选择性。值得注意的是,该反应具有反应条件温和、底物范围广泛以及良好的官能团相容性等特点。此外,大规模合成、减少催化剂负载量实验以及手性邻苯二甲酸酯前药的合成也强调了该方法的实用性。
更新日期:2024-05-27
中文翻译:

手性异硫脲催化 3-羟基苯酞的酰化动态动力学拆分用于对映选择性合成邻苯二甲酸酯
邻苯二甲酸酯作为核心结构广泛存在于各种天然产物和药物分子中,表现出多种生物活性。我们在此报告了通过手性异硫脲(ITU)催化的不对称酰化对3-羟基苯酞进行高效动态动力学拆分,促进了各种手性苯酞酯的有效合成,具有良好的产率和对映选择性。值得注意的是,该反应具有反应条件温和、底物范围广泛以及良好的官能团相容性等特点。此外,大规模合成、减少催化剂负载量实验以及手性邻苯二甲酸酯前药的合成也强调了该方法的实用性。

































 京公网安备 11010802027423号
京公网安备 11010802027423号