当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Reaction of 1,3,5-Triazinanes with Phosphoryl Diazomethanes: Access to 5-Phosphoryl-1,2,3,4-tetrahydropyrimidines
Organic Letters ( IF 4.9 ) Pub Date : 2024-04-17 , DOI: 10.1021/acs.orglett.4c00881 Wenlai Xie 1 , Chengzhuo Wang 1 , Jiaxi Xu 1
Organic Letters ( IF 4.9 ) Pub Date : 2024-04-17 , DOI: 10.1021/acs.orglett.4c00881 Wenlai Xie 1 , Chengzhuo Wang 1 , Jiaxi Xu 1
Affiliation
|
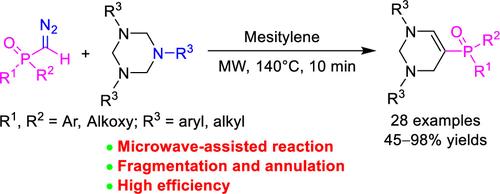
Conversion of 1,3,5-triazinanes into 5-phosporyl-1,2,3,4-tetrahydropyrimidines is achieved efficiently through the microwave-assisted reaction with phosphoryl diazomethanes. Both trialkyl and triaryl 1,3,5-triazinanes were converted by diazomethyldiarylphosphine oxides, dialkyl diazomethylphosphonates, and alkyl diazomethyl(aryl)phosphinates and functionalized simultaneously in good to excellent yields. The reaction is a sequence of 1,3,5-triazinane fragmentation, tandem nucleophilic addition of the generated formaldimines and phosphoryl diazomethanes, and final N,N-acetalization.
中文翻译:

1,3,5-三嗪与磷酰重氮甲烷的反应:获得 5-磷酰-1,2,3,4-四氢嘧啶
通过与磷酰重氮甲烷的微波辅助反应,可以有效地将 1,3,5-三嗪转化为 5-磷酰基-1,2,3,4-四氢嘧啶。三烷基和三芳基1,3,5-三嗪烷均可通过重氮甲基二芳基氧化膦、重氮甲基膦酸二烷基酯和重氮甲基(芳基)次膦酸烷基酯转化并同时官能化,收率良好至优异。该反应是一系列 1,3,5-三嗪烷断裂、生成的福尔马亚胺和磷酰重氮甲烷的串联亲核加成,以及最终的N , N-缩醛化。
更新日期:2024-04-17
中文翻译:

1,3,5-三嗪与磷酰重氮甲烷的反应:获得 5-磷酰-1,2,3,4-四氢嘧啶
通过与磷酰重氮甲烷的微波辅助反应,可以有效地将 1,3,5-三嗪转化为 5-磷酰基-1,2,3,4-四氢嘧啶。三烷基和三芳基1,3,5-三嗪烷均可通过重氮甲基二芳基氧化膦、重氮甲基膦酸二烷基酯和重氮甲基(芳基)次膦酸烷基酯转化并同时官能化,收率良好至优异。该反应是一系列 1,3,5-三嗪烷断裂、生成的福尔马亚胺和磷酰重氮甲烷的串联亲核加成,以及最终的N , N-缩醛化。






























 京公网安备 11010802027423号
京公网安备 11010802027423号