当前位置:
X-MOL 学术
›
Curr. Res. Food Sci.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
The evaluation of catechins reducing heterocyclic aromatic amine formation: Structure-activity relationship and mechanism speculation
Current Research in Food Science ( IF 6.2 ) Pub Date : 2024-03-30 , DOI: 10.1016/j.crfs.2024.100727 Ruiwei Xie , Haolin Zhang , Xiaomei Lv , Qiuyi Lin , Bing-Huei Chen , Yu-Wen Lai , Lei Chen , Hui Teng , Hui Cao
Current Research in Food Science ( IF 6.2 ) Pub Date : 2024-03-30 , DOI: 10.1016/j.crfs.2024.100727 Ruiwei Xie , Haolin Zhang , Xiaomei Lv , Qiuyi Lin , Bing-Huei Chen , Yu-Wen Lai , Lei Chen , Hui Teng , Hui Cao
|
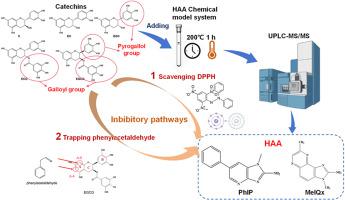
The favorable inhibitory effect of tea polyphenols on heterocyclic aromatic amines (HAAs) has been confirmed in many past studies. The objective of this study was to investigate the structure-activity relationship of catechins that act as inhibitors of HAA formation in chemical models. Two kinds of quantitative structure-activity relationship models for catechin-inhibiting-HAA were established. We chose two kinds of HAAs including 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) and 2-amino-3,8-dimethylimidazo[4,5-f]quinoxaline (MeIQx), and five catechins including epigallocatechin gallate (EGCG), epicatechin gallate (ECG), epigallocatechin (EGC), epicatechin (EC), and catechin (C). The inhibitory effect of five catechins were in the following order: EGCG > ECG > EGC > C > EC. Thereinto, EGCG and ECG showed dramatically better inhibition on the formation of PhIP and MeIQx, especially EGCG. Further, the mechanisms of catechin-inhibiting-HAA were speculated by correlation analysis. The free radical-scavenging ability was predicted to be the most relevant to the inhibitory effect of ECG, EGC, EC and C on HAAs. Differently, the phenylacetaldehyde-trapping ability might be the more important mechanism of EGCG inhibiting PhIP in chemical model system. This study may bring a broader idea for controlling the formation of HAAs according to the structure of catechins.
中文翻译:

儿茶素减少杂环芳香胺形成的评价:构效关系和机制推测
茶多酚对杂环芳香胺(HAAs)的良好抑制作用已在过去的许多研究中得到证实。本研究的目的是研究儿茶素在化学模型中作为 HAA 形成抑制剂的结构-活性关系。建立了儿茶素抑制HAA的两种定量构效关系模型。我们选择了两种HAA,包括2-氨基-1-甲基-6-苯基咪唑并[4,5-b]吡啶(PhIP)和2-氨基-3,8-二甲基咪唑[4,5-f]喹喔啉(MeIQx)和五种儿茶素,包括表没食子儿茶素没食子酸酯(EGCG)、表儿茶素没食子酸酯(ECG)、表没食子儿茶素(EGC)、表儿茶素(EC)和儿茶素(C)。 5种儿茶素的抑制作用顺序为:EGCG>ECG>EGC>C>EC。其中,EGCG和ECG对PhIP和MeIQx的形成表现出显着更好的抑制作用,尤其是EGCG。此外,通过相关分析推测儿茶素抑制HAA的机制。预计自由基清除能力与 ECG、EGC、EC 和 C 对 HAA 的抑制作用最相关。不同的是,苯乙醛捕获能力可能是化学模型系统中EGCG抑制PhIP的更重要机制。这项研究可能为根据儿茶素的结构控制HAA的形成带来更广泛的思路。
更新日期:2024-03-30
中文翻译:

儿茶素减少杂环芳香胺形成的评价:构效关系和机制推测
茶多酚对杂环芳香胺(HAAs)的良好抑制作用已在过去的许多研究中得到证实。本研究的目的是研究儿茶素在化学模型中作为 HAA 形成抑制剂的结构-活性关系。建立了儿茶素抑制HAA的两种定量构效关系模型。我们选择了两种HAA,包括2-氨基-1-甲基-6-苯基咪唑并[4,5-b]吡啶(PhIP)和2-氨基-3,8-二甲基咪唑[4,5-f]喹喔啉(MeIQx)和五种儿茶素,包括表没食子儿茶素没食子酸酯(EGCG)、表儿茶素没食子酸酯(ECG)、表没食子儿茶素(EGC)、表儿茶素(EC)和儿茶素(C)。 5种儿茶素的抑制作用顺序为:EGCG>ECG>EGC>C>EC。其中,EGCG和ECG对PhIP和MeIQx的形成表现出显着更好的抑制作用,尤其是EGCG。此外,通过相关分析推测儿茶素抑制HAA的机制。预计自由基清除能力与 ECG、EGC、EC 和 C 对 HAA 的抑制作用最相关。不同的是,苯乙醛捕获能力可能是化学模型系统中EGCG抑制PhIP的更重要机制。这项研究可能为根据儿茶素的结构控制HAA的形成带来更广泛的思路。











































 京公网安备 11010802027423号
京公网安备 11010802027423号