当前位置:
X-MOL 学术
›
J. Med. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Discovery of 2,3-Dihydro[1,4]dioxino[2,3-g]benzofuran Derivatives as Protease Activated Receptor 4 (PAR4) Antagonists with Potent Antiplatelet Aggregation Activity and Low Bleeding Tendency
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2024-03-29 , DOI: 10.1021/acs.jmedchem.3c02099 Panpan Chen 1, 2 , Cai Chen 1 , Yizheng Zheng 3 , Fangjun Chen 1 , Zhaojun Liu 3 , Shenhong Ren 3 , Hangyu Song 1 , Tongdan Liu 3 , Zhipeng Lu 2 , Hongbin Sun 1, 4 , Yi Kong 3 , Haoliang Yuan 1, 4
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2024-03-29 , DOI: 10.1021/acs.jmedchem.3c02099 Panpan Chen 1, 2 , Cai Chen 1 , Yizheng Zheng 3 , Fangjun Chen 1 , Zhaojun Liu 3 , Shenhong Ren 3 , Hangyu Song 1 , Tongdan Liu 3 , Zhipeng Lu 2 , Hongbin Sun 1, 4 , Yi Kong 3 , Haoliang Yuan 1, 4
Affiliation
|
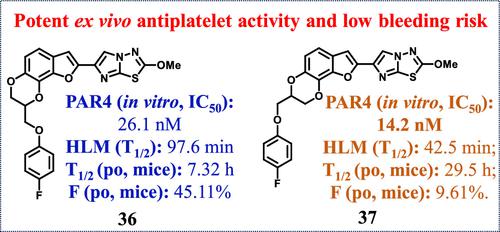
Patients with arterial embolic disease have benefited greatly from antiplatelet therapy. However, hemorrhage risk of antiplatelet agents cannot be ignored. Herein, we describe the discovery of 2,3-dihydro[1,4]dioxino[2,3-g]benzofuran compounds as novel PAR4 antagonists. Notably, the isomers 36 and 37 with the chemotype of phenoxyl methylene substituted on the 2,3-dihydro-1,4-dioxine ring exhibited potent in vitro antiplatelet activity (IC50 = 26.13 nM for 36 and 14.26 nM for 37) and significantly improved metabolic stability in human liver microsomes (T1/2 = 97.6 min for 36 and 11.1 min for BMS-986120). 36 also displayed good oral PK profiles (mice: T1/2 = 7.32 h and F = 45.11%). Both of them showed overall potent ex vivo antiplatelet activity at concentrations of 6 and 12 mg/kg, with no impact on the coagulation system and low bleeding liability. Our work will facilitate development of novel PAR4 antagonists as a safer therapeutic option for arterial embolism.
中文翻译:

发现 2,3-二氢[1,4]二恶英[2,3-g]苯并呋喃衍生物作为蛋白酶激活受体 4 (PAR4) 拮抗剂,具有有效的抗血小板聚集活性和低出血倾向
动脉栓塞性疾病患者从抗血小板治疗中获益匪浅。然而,抗血小板药物的出血风险也不容忽视。在此,我们描述了 2,3-二氢[1,4]二恶英[2,3- g ]苯并呋喃化合物作为新型 PAR4 拮抗剂的发现。值得注意的是,化学型为苯氧基亚甲基在 2,3-二氢-1,4-二恶英环上取代的异构体36和37表现出有效的体外抗血小板活性( 36的 IC 50 = 26.13 nM, 37的 IC 50 = 14.26 nM),并且显着改善人肝微粒体的代谢稳定性(BMS-986120 36时T 1/2 = 97.6 分钟,11.1 分钟)。 36还表现出良好的口服 PK 特征(小鼠: T 1/2 = 7.32 h 和F = 45.11%)。两者在浓度为 6 和 12 mg/kg 时均表现出有效的体外抗血小板活性,对凝血系统没有影响且出血倾向低。我们的工作将促进新型 PAR4 拮抗剂的开发,作为动脉栓塞更安全的治疗选择。
更新日期:2024-03-29
中文翻译:

发现 2,3-二氢[1,4]二恶英[2,3-g]苯并呋喃衍生物作为蛋白酶激活受体 4 (PAR4) 拮抗剂,具有有效的抗血小板聚集活性和低出血倾向
动脉栓塞性疾病患者从抗血小板治疗中获益匪浅。然而,抗血小板药物的出血风险也不容忽视。在此,我们描述了 2,3-二氢[1,4]二恶英[2,3- g ]苯并呋喃化合物作为新型 PAR4 拮抗剂的发现。值得注意的是,化学型为苯氧基亚甲基在 2,3-二氢-1,4-二恶英环上取代的异构体36和37表现出有效的体外抗血小板活性( 36的 IC 50 = 26.13 nM, 37的 IC 50 = 14.26 nM),并且显着改善人肝微粒体的代谢稳定性(BMS-986120 36时T 1/2 = 97.6 分钟,11.1 分钟)。 36还表现出良好的口服 PK 特征(小鼠: T 1/2 = 7.32 h 和F = 45.11%)。两者在浓度为 6 和 12 mg/kg 时均表现出有效的体外抗血小板活性,对凝血系统没有影响且出血倾向低。我们的工作将促进新型 PAR4 拮抗剂的开发,作为动脉栓塞更安全的治疗选择。






























 京公网安备 11010802027423号
京公网安备 11010802027423号