当前位置:
X-MOL 学术
›
Biopharm. Drug Dispos.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Characterization of AST‐001 non‐clinical pharmacokinetics: A novel selective AKR1C3‐activated prodrug in mice, rats, and cynomolgus monkeys
Biopharmaceutics & Drug Disposition ( IF 1.7 ) Pub Date : 2024-03-16 , DOI: 10.1002/bdd.2385
Teng Meng 1 , Donald Jung 1 , Xiao-Hong Cai 1 , Zhao-Qiang Lu 1 , Ji-Bing Yu 1 , Tian-Yang Qi 1 , Fan-Ying Meng 1 , Mei-Zhen Ruan 1 , Jian-Xin Duan 1
Biopharmaceutics & Drug Disposition ( IF 1.7 ) Pub Date : 2024-03-16 , DOI: 10.1002/bdd.2385
Teng Meng 1 , Donald Jung 1 , Xiao-Hong Cai 1 , Zhao-Qiang Lu 1 , Ji-Bing Yu 1 , Tian-Yang Qi 1 , Fan-Ying Meng 1 , Mei-Zhen Ruan 1 , Jian-Xin Duan 1
Affiliation
|
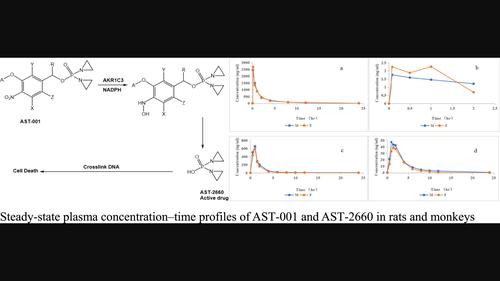
AST‐001 is a chemically synthesized inactive nitrogen mustard prodrug that is selectively cleaved to a cytotoxic aziridine (AST‐2660) via aldo‐keto reductase family 1 member C3 (AKR1C3). The purpose of this study was to investigate the pharmacokinetics and tissue distribution of the prodrug, AST‐001, and its active metabolite, AST‐2660, in mice, rats, and monkeys. After single and once daily intravenous bolus doses of 1.5, 4.5, and 13.5 mg/kg AST‐001 to Sprague‐Dawley rats and once daily 1 h intravenous infusions of 0.5, 1.5, and 4.5 mg/kg AST‐001 to cynomolgus monkeys, AST‐001 exhibited dose‐dependent pharmacokinetics and reached peak plasma levels at the end of the infusion. No significant accumulation and gender differences were observed after 7 days of repeated dosing. In rats, the half‐life of AST‐001 was dose independent and ranged from 4.89 to 5.75 h. In cynomolgus monkeys, the half‐life of AST‐001 was from 1.66 to 5.56 h and increased with dose. In tissue distribution studies conducted in Sprague‐Dawley rats and in liver cancer PDX models in female athymic nude mice implanted with LI6643 or LI6280 HepG2‐GFP tumor fragments, AST‐001 was extensively distributed to selected tissues. Following a single intravenous dose, AST‐001 was not excreted primarily as the prodrug, AST‐001 or the metabolite AST‐2660 in the urine, feces, and bile. A comprehensive analysis of the preclinical data and inter‐species allometric scaling were used to estimate the pharmacokinetic parameters of AST‐001 in humans and led to the recommendation of a starting dose of 5 mg/m2 in the first‐in‐human dose escalation study.
中文翻译:

AST-001 非临床药代动力学表征:一种新型选择性 AKR1C3 激活前药在小鼠、大鼠和食蟹猴中的作用
AST-001 是一种化学合成的无活性氮芥前药,可通过醛酮还原酶家族 1 成员 C3 (AKR1C3) 选择性裂解为细胞毒性氮丙啶 (AST-2660)。本研究的目的是研究前药 AST-001 及其活性代谢物 AST-2660 在小鼠、大鼠和猴子中的药代动力学和组织分布。对 Sprague-Dawley 大鼠单次和每日一次静脉推注剂量 1.5、4.5 和 13.5 mg/kg AST-001 以及对食蟹猴每日一次 1 小时静脉输注 0.5、1.5 和 4.5 mg/kg AST-001 后, AST-001 表现出剂量依赖性药代动力学,并在输注结束时达到峰值血浆水平。重复给药7天后未观察到明显的蓄积和性别差异。在大鼠中,AST-001 的半衰期与剂量无关,范围为 4.89 至 5.75 小时。在食蟹猴中,AST-001 的半衰期为 1.66 至 5.56 小时,并随着剂量的增加而增加。在 Sprague-Dawley 大鼠和植入 LI6643 或 LI6280 HepG2-GFP 肿瘤片段的雌性无胸腺裸鼠的肝癌 PDX 模型中进行的组织分布研究中,AST-001 广泛分布到选定的组织中。单次静脉给药后,AST-001 主要不作为前药 AST-001 或代谢物 AST-2660 通过尿液、粪便和胆汁排出。对临床前数据和种间异速生长标度的综合分析用于估计 AST-001 在人体中的药代动力学参数,并建议起始剂量为 5 mg/m2 2在首次人体剂量递增研究中。
更新日期:2024-03-16
中文翻译:

AST-001 非临床药代动力学表征:一种新型选择性 AKR1C3 激活前药在小鼠、大鼠和食蟹猴中的作用
AST-001 是一种化学合成的无活性氮芥前药,可通过醛酮还原酶家族 1 成员 C3 (AKR1C3) 选择性裂解为细胞毒性氮丙啶 (AST-2660)。本研究的目的是研究前药 AST-001 及其活性代谢物 AST-2660 在小鼠、大鼠和猴子中的药代动力学和组织分布。对 Sprague-Dawley 大鼠单次和每日一次静脉推注剂量 1.5、4.5 和 13.5 mg/kg AST-001 以及对食蟹猴每日一次 1 小时静脉输注 0.5、1.5 和 4.5 mg/kg AST-001 后, AST-001 表现出剂量依赖性药代动力学,并在输注结束时达到峰值血浆水平。重复给药7天后未观察到明显的蓄积和性别差异。在大鼠中,AST-001 的半衰期与剂量无关,范围为 4.89 至 5.75 小时。在食蟹猴中,AST-001 的半衰期为 1.66 至 5.56 小时,并随着剂量的增加而增加。在 Sprague-Dawley 大鼠和植入 LI6643 或 LI6280 HepG2-GFP 肿瘤片段的雌性无胸腺裸鼠的肝癌 PDX 模型中进行的组织分布研究中,AST-001 广泛分布到选定的组织中。单次静脉给药后,AST-001 主要不作为前药 AST-001 或代谢物 AST-2660 通过尿液、粪便和胆汁排出。对临床前数据和种间异速生长标度的综合分析用于估计 AST-001 在人体中的药代动力学参数,并建议起始剂量为 5 mg/m2 2在首次人体剂量递增研究中。

































 京公网安备 11010802027423号
京公网安备 11010802027423号