当前位置:
X-MOL 学术
›
Exp. Neurol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
BMSC-Exosomes attenuate ALP dysfunction by restoring lysosomal function via the mTOR/TFEB Axis to reduce cerebral ischemia-reperfusion injury
Experimental Neurology ( IF 4.6 ) Pub Date : 2024-02-23 , DOI: 10.1016/j.expneurol.2024.114726
Haining Liu 1 , Chen Li 2 , Xiaofeng Zhang 1 , Hui Chen 1 , Qi Zhang 1 , Yuting Zeng 1 , Shuqi Zheng 1 , Jihua Zou 3 , Yijin Zhao 1 , Xiaoyan Zheng 4 , Guozhi Huang 1 , Qing Zeng 1
Experimental Neurology ( IF 4.6 ) Pub Date : 2024-02-23 , DOI: 10.1016/j.expneurol.2024.114726
Haining Liu 1 , Chen Li 2 , Xiaofeng Zhang 1 , Hui Chen 1 , Qi Zhang 1 , Yuting Zeng 1 , Shuqi Zheng 1 , Jihua Zou 3 , Yijin Zhao 1 , Xiaoyan Zheng 4 , Guozhi Huang 1 , Qing Zeng 1
Affiliation
|
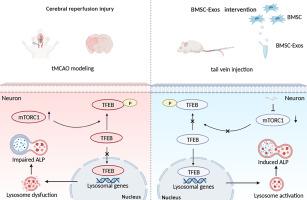
The complex pathophysiological changes following cerebral ischemia-reperfusion injury (CIRI) include the accumulation of defective proteins and damaged organelles, which cause massive neuron demise. To preserve cellular homeostasis, the autophagy-lysosomal pathway (ALP) is crucial for neurons to dispose of these substances. Many studies have shown that bone mesenchymal stem cell exosomes (BMSC-Exos) can reduce CIRI. However, the specific mechanisms have not been well elucidated, a fact that limits its widespread clinical use. This study aimed to clarify whether BMSC-Exos could attenuate ALP dysfunction by restoring lysosomal function after CIRI via inhibiting mTOR and then activating TFEB nucleus translocation. In this study, Flow cytometry, Nanoparticle tracking analysis (NTA), Transmission electron microscope (TEM), and Western blot were used to identify the BMSCs and BMSC-Exos used in this experiment as conforming to the requirements. In vivo experiments, SD rats were modeled with temporary middle cerebral artery occlusion (tMCAO), and BMSC-Exos was injected into the tail vein 2 h after modeling. Triphenyl tetrazolium chloride (TTC) staining, modified neurological severity scores (mNSS), corner turn test, and rotating rod test were used to detect neurological deficits in rats after BMSC-Exos intervention. Western blot and Immunofluorescence were used to detect ALP, transcription factor EB(TFEB) nucleus translocation, and mammalian target of rapamycin (mTOR) change at different time points after modeling and after BMSC-Exos intervention. In vitro experiments, pheochromocytoma cells (PC12) cells were subjected to oxygen-glucose deprivation and reperfusion (OGD/R) modeling to mimic CIRI, and were respectively intervened with BMSC-Exos, BMSC-Exos + MHY 1485 (the mTOR agonist), Rapamycin (the mTOR inhibitor). CCK8, Western blot, and Immunofluorescence were used to detect PC12 cell survival, TFEB nucleus translocation, and cathepsin B(CTSB) Immunofluorescence intensity. We found that ALP dysfunction occurred 72 h after tMCAO, and BMSC-Exos can attenuate ALP dysfunction by restoring lysosomal function. Next, we examined TFEB nucleus translocation and the expression of mTOR, a key regulator of translocation. We found that BMSC-Exos could inhibit mTOR and activate TFEB nucleus translocation. Additional in vitro tests revealed that BMSC-Exos could increase PC12 cell survival after OGD/R, activating TFEB nucleus translocation and enhancing the fluorescence intensity of CTSB, which in turn could be reversed by the mTOR agonist, MHY1485. This effect was similar to another mTOR inhibitor, Rapamycin. BMSC-Exos could attenuate ALP dysfunction by restoring lysosomal function after CIRI by inhibiting mTOR and then promoting TFEB nucleus translocation.
中文翻译:

BMSC-外泌体通过 mTOR/TFEB 轴恢复溶酶体功能,从而减轻 ALP 功能障碍,从而减少脑缺血再灌注损伤
脑缺血再灌注损伤(CIRI)后复杂的病理生理变化包括缺陷蛋白质和受损细胞器的积累,从而导致大量神经元死亡。为了保持细胞稳态,自噬-溶酶体途径 (ALP) 对于神经元处理这些物质至关重要。许多研究表明,骨间充质干细胞外泌体(BMSC-Exos)可以降低CIRI。然而,其具体机制尚未得到很好的阐明,这限制了其广泛的临床应用。本研究旨在阐明 BMSC-Exos 是否可以通过抑制 mTOR 然后激活 TFEB 核易位来恢复 CIRI 后溶酶体功能,从而减轻 ALP 功能障碍。本研究通过流式细胞术、纳米粒子追踪分析(NTA)、透射电镜(TEM)、Western blot等方法鉴定本实验使用的BMSCs和BMSC-Exos符合要求。体内实验中,SD大鼠采用大脑中动脉暂时闭塞(tMCAO)造模,造模后2 h尾静脉注射BMSC-Exos。采用氯化三苯基四唑(TTC)染色、改良神经严重程度评分(mNSS)、转角试验和旋转棒试验检测BMSC-Exos干预后大鼠的神经功能缺损。采用Western blot和免疫荧光检测造模后和BMSC-Exos干预后不同时间点ALP、转录因子EB(TFEB)核转位以及哺乳动物雷帕霉素靶蛋白(mTOR)的变化。 体外实验中,对嗜铬细胞瘤细胞(PC12)细胞进行氧糖剥夺再灌注(OGD/R)建模,模拟CIRI,并分别用BMSC-Exos、BMSC-Exos + MHY 1485(mTOR激动剂)、雷帕霉素(mTOR 抑制剂)。采用CCK8、Western blot、免疫荧光检测PC12细胞存活、TFEB核转位、组织蛋白酶B(CTSB)免疫荧光强度。我们发现ALP功能障碍发生在tMCAO后72小时,BMSC-Exos可以通过恢复溶酶体功能来减轻ALP功能障碍。接下来,我们检查了 TFEB 核易位和易位关键调节因子 mTOR 的表达。我们发现BMSC-Exos可以抑制mTOR并激活TFEB核易位。其他体外测试表明,BMSC-Exos 可以增加 OGD/R 后 PC12 细胞的存活率,激活 TFEB 核易位并增强 CTSB 的荧光强度,而这反过来又可以被 mTOR 激动剂 MHY1485 逆转。这种效果与另一种 mTOR 抑制剂雷帕霉素类似。 BMSC-Exos 可以通过抑制 mTOR 然后促进 TFEB 核易位,恢复 CIRI 后的溶酶体功能,从而减轻 ALP 功能障碍。
更新日期:2024-02-23
中文翻译:

BMSC-外泌体通过 mTOR/TFEB 轴恢复溶酶体功能,从而减轻 ALP 功能障碍,从而减少脑缺血再灌注损伤
脑缺血再灌注损伤(CIRI)后复杂的病理生理变化包括缺陷蛋白质和受损细胞器的积累,从而导致大量神经元死亡。为了保持细胞稳态,自噬-溶酶体途径 (ALP) 对于神经元处理这些物质至关重要。许多研究表明,骨间充质干细胞外泌体(BMSC-Exos)可以降低CIRI。然而,其具体机制尚未得到很好的阐明,这限制了其广泛的临床应用。本研究旨在阐明 BMSC-Exos 是否可以通过抑制 mTOR 然后激活 TFEB 核易位来恢复 CIRI 后溶酶体功能,从而减轻 ALP 功能障碍。本研究通过流式细胞术、纳米粒子追踪分析(NTA)、透射电镜(TEM)、Western blot等方法鉴定本实验使用的BMSCs和BMSC-Exos符合要求。体内实验中,SD大鼠采用大脑中动脉暂时闭塞(tMCAO)造模,造模后2 h尾静脉注射BMSC-Exos。采用氯化三苯基四唑(TTC)染色、改良神经严重程度评分(mNSS)、转角试验和旋转棒试验检测BMSC-Exos干预后大鼠的神经功能缺损。采用Western blot和免疫荧光检测造模后和BMSC-Exos干预后不同时间点ALP、转录因子EB(TFEB)核转位以及哺乳动物雷帕霉素靶蛋白(mTOR)的变化。 体外实验中,对嗜铬细胞瘤细胞(PC12)细胞进行氧糖剥夺再灌注(OGD/R)建模,模拟CIRI,并分别用BMSC-Exos、BMSC-Exos + MHY 1485(mTOR激动剂)、雷帕霉素(mTOR 抑制剂)。采用CCK8、Western blot、免疫荧光检测PC12细胞存活、TFEB核转位、组织蛋白酶B(CTSB)免疫荧光强度。我们发现ALP功能障碍发生在tMCAO后72小时,BMSC-Exos可以通过恢复溶酶体功能来减轻ALP功能障碍。接下来,我们检查了 TFEB 核易位和易位关键调节因子 mTOR 的表达。我们发现BMSC-Exos可以抑制mTOR并激活TFEB核易位。其他体外测试表明,BMSC-Exos 可以增加 OGD/R 后 PC12 细胞的存活率,激活 TFEB 核易位并增强 CTSB 的荧光强度,而这反过来又可以被 mTOR 激动剂 MHY1485 逆转。这种效果与另一种 mTOR 抑制剂雷帕霉素类似。 BMSC-Exos 可以通过抑制 mTOR 然后促进 TFEB 核易位,恢复 CIRI 后的溶酶体功能,从而减轻 ALP 功能障碍。

































 京公网安备 11010802027423号
京公网安备 11010802027423号