当前位置:
X-MOL 学术
›
J. Clean. Prod.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Efficient lithium recovery from electrolytic aluminum slag via an environmentally friendly process: Leaching behavior and mechanism
Journal of Cleaner Production ( IF 9.7 ) Pub Date : 2024-01-18 , DOI: 10.1016/j.jclepro.2024.140800
Li Cui , Weihong Wang , Xi Chao , Jianming Gao , Fangqin Cheng
Journal of Cleaner Production ( IF 9.7 ) Pub Date : 2024-01-18 , DOI: 10.1016/j.jclepro.2024.140800
Li Cui , Weihong Wang , Xi Chao , Jianming Gao , Fangqin Cheng
|
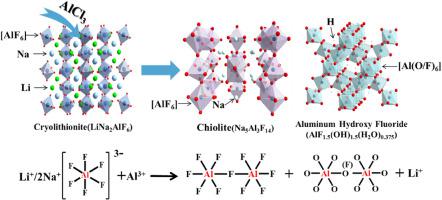
The electrolytic aluminum industry generates a large amount lithium (Li)-containing electrolytic aluminum slag (EAS) annually, and this can result in severe environmental pollution and wasting of Li resources. This paper proposes a method for recovering Li from the slag using AlCl as the leaching agent. The effects of the leaching temperature, AlCl concentration, pH, and solid-to-liquid ratio (S/L) on the leaching efficiency of Li were systematically examined. The structural transformation during the Li leaching process was revealed by SEM, XRD, Al NMR, and XPS analyses. The results indicate that up to 88.3% of Li can be leached out using 0.85 M of AlCl with an S/L of 1:3 at a leaching temperature of 95 °C and pH of 0.5. The addition of AlCl played an essential role in the decomposition of cryolite/cryolithionite, ultimately resulting in Al–F fracture and Li release in the leaching solution. Moreover, Al enters the interior of the slag to participate in the reconstruction of the chiolite with a low NaF/AlF ratio, and reacts with OH ionized from HO to form hydroxyl aluminum fluoride species. Fluorine ions were solidified in the leaching residues for subsequent cryolite regeneration. This study not only provides an environmentally friendly process for resource utilization from EAS but also reduces the environmental threat caused by the storage of the hazardous waste.
中文翻译:

通过环保工艺从电解铝渣中高效回收锂:浸出行为和机制
电解铝工业每年产生大量的含锂电解铝渣(EAS),这会造成严重的环境污染和锂资源的浪费。本文提出了一种以AlCl为浸出剂从炉渣中回收Li的方法。系统地考察了浸出温度、AlCl浓度、pH和固液比(S/L)对锂浸出效率的影响。通过 SEM、XRD、Al NMR 和 XPS 分析揭示了锂浸出过程中的结构转变。结果表明,在浸出温度为 95 °C、pH 值为 0.5 时,使用 0.85 M AlCl、S/L 为 1:3 可以浸出高达 88.3% 的锂。 AlCl的添加在冰晶石/冰锂石的分解中发挥了重要作用,最终导致浸出液中Al-F断裂和Li释放。此外,Al进入炉渣内部参与低NaF/AlF比的硅沸石的重构,并与H2O电离的OH反应形成羟基氟化铝物质。氟离子在浸出残渣中固化,用于随后的冰晶石再生。这项研究不仅为EAS的资源利用提供了一种环保的工艺,而且减少了危险废物储存造成的环境威胁。
更新日期:2024-01-18
中文翻译:

通过环保工艺从电解铝渣中高效回收锂:浸出行为和机制
电解铝工业每年产生大量的含锂电解铝渣(EAS),这会造成严重的环境污染和锂资源的浪费。本文提出了一种以AlCl为浸出剂从炉渣中回收Li的方法。系统地考察了浸出温度、AlCl浓度、pH和固液比(S/L)对锂浸出效率的影响。通过 SEM、XRD、Al NMR 和 XPS 分析揭示了锂浸出过程中的结构转变。结果表明,在浸出温度为 95 °C、pH 值为 0.5 时,使用 0.85 M AlCl、S/L 为 1:3 可以浸出高达 88.3% 的锂。 AlCl的添加在冰晶石/冰锂石的分解中发挥了重要作用,最终导致浸出液中Al-F断裂和Li释放。此外,Al进入炉渣内部参与低NaF/AlF比的硅沸石的重构,并与H2O电离的OH反应形成羟基氟化铝物质。氟离子在浸出残渣中固化,用于随后的冰晶石再生。这项研究不仅为EAS的资源利用提供了一种环保的工艺,而且减少了危险废物储存造成的环境威胁。

































 京公网安备 11010802027423号
京公网安备 11010802027423号