当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Mechanochemistry of Pterodactylane
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2023-12-22 , DOI: 10.1021/jacs.3c11293
Matías Horst 1 , Jan Meisner 1, 2 , Jinghui Yang 1 , Tatiana B Kouznetsova 3 , Stephen L Craig 3 , Todd J Martínez 1, 4 , Yan Xia 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2023-12-22 , DOI: 10.1021/jacs.3c11293
Matías Horst 1 , Jan Meisner 1, 2 , Jinghui Yang 1 , Tatiana B Kouznetsova 3 , Stephen L Craig 3 , Todd J Martínez 1, 4 , Yan Xia 1
Affiliation
|
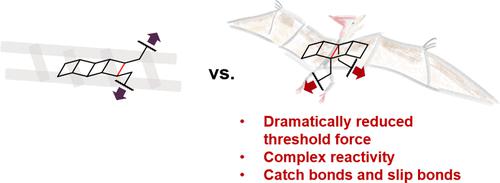
Pterodactylane is a [4]-ladderane with substituents on the central rung. Comparing the mechanochemistry of the [4]-ladderane structure when pulled from the central rung versus the end rung revealed a striking difference in the threshold force of mechanoactivation: the threshold force is dramatically lowered from 1.9 nN when pulled on the end rung to 0.7 nN when pulled on the central rung. We investigated the bicyclic products formed from the mechanochemical activation of pterodactylane experimentally and computationally, which are distinct from the mechanochemical products of ladderanes being activated from the end rung. We compared the products of pterodactylane’s mechanochemical and thermal activation to reveal differences and similarities in the mechanochemical and thermal pathways of pterodactylane transformation. Interestingly, we also discovered the presence of elementary steps that are accelerated or suppressed by force within the same mechanochemical reaction of pterodactylane, suggesting rich mechanochemical manifolds of multicyclic structures. We rationalized the greatly enhanced mechanochemical reactivity of the central rung of pterodactylane and discovered force-free ground state bond length to be a good low-cost predictor of the threshold force for cyclobutane-based mechanophores. These findings advance our understanding of mechanochemical reactivities and pathways, and they will guide future designs of mechanophores with low threshold forces to facilitate their applications in force-responsive materials.
中文翻译:

翼手龙的力学化学
翼手龙是一种在中央横档带有取代基的[4]-梯烷。比较从中央横档与末端横档拉动时的 [4]-ladderane 结构的机械化学,揭示了机械激活阈值力的显着差异:阈值力从从末端横档拉动时的 1.9 nN 急剧降低到 0.7 nN当拉动中央横档时。我们通过实验和计算研究了翼手龙机械化学活化形成的双环产物,这与从末端梯级活化的梯烷机械化学产物不同。我们比较了翼手龙的机械化学和热活化产物,以揭示翼手龙转化的机械化学和热途径的差异和相似之处。有趣的是,我们还发现在翼手龙的同一机械化学反应中存在被力加速或抑制的基本步骤,这表明多环结构具有丰富的机械化学流形。我们合理化了翼手龙中央梯级大大增强的机械化学反应性,并发现无力基态键长是基于环丁烷的机械力团阈值力的良好低成本预测因子。这些发现增进了我们对机械化学反应性和途径的理解,并将指导未来具有低阈值力的机械载体的设计,以促进其在力响应材料中的应用。
更新日期:2023-12-22
中文翻译:

翼手龙的力学化学
翼手龙是一种在中央横档带有取代基的[4]-梯烷。比较从中央横档与末端横档拉动时的 [4]-ladderane 结构的机械化学,揭示了机械激活阈值力的显着差异:阈值力从从末端横档拉动时的 1.9 nN 急剧降低到 0.7 nN当拉动中央横档时。我们通过实验和计算研究了翼手龙机械化学活化形成的双环产物,这与从末端梯级活化的梯烷机械化学产物不同。我们比较了翼手龙的机械化学和热活化产物,以揭示翼手龙转化的机械化学和热途径的差异和相似之处。有趣的是,我们还发现在翼手龙的同一机械化学反应中存在被力加速或抑制的基本步骤,这表明多环结构具有丰富的机械化学流形。我们合理化了翼手龙中央梯级大大增强的机械化学反应性,并发现无力基态键长是基于环丁烷的机械力团阈值力的良好低成本预测因子。这些发现增进了我们对机械化学反应性和途径的理解,并将指导未来具有低阈值力的机械载体的设计,以促进其在力响应材料中的应用。

































 京公网安备 11010802027423号
京公网安备 11010802027423号