Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
介孔碳气凝胶浸渍支化聚乙烯亚胺的结构-功能关系:深入的热重分析
Langmuir
(
IF
3.7
)
Pub Date : 2023-11-17
, DOI:
10.1021/acs.langmuir.3c02043
Alejandra A Martinez
1,
2
,
Pierre P Arneodo Larochette
1,
3
,
Fabiana C Gennari
1,
3
,
Aurelien Gasnier
1,
2
Affiliation
- Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) and Centro Atómico Bariloche (CNEA), S. C. de Bariloche, Río Negro R8402AGP, Argentina.
- Instituto de Nanociencia y Nanotecnología, S. C. de Bariloche, Río Negro R8402AGP, Argentina.
- Instituto Balseiro, Universidad Nacional de Cuyo, S. C. de Bariloche, Río Negro R8402AGP, Argentina.
我们对聚乙烯亚胺 (PEI) 浸渍的间苯二酚-甲醛 (RF) 气凝胶进行了全面的热重分析 (TGA)。虽然许多研究都集中在 PEI 浸渍的 SBA 上,但射频材料尽管具有兴趣和特殊性,但对其研究较少。由于大多数有关 PEI 浸渍多孔材料的文章都遵循 SBA 定义的典型实验方法,因此 RF-PEI 材料的特殊性可能仍然被忽视。非等温 TGA 方案的设计,以氮等温线完成,基于基质的系统填充,提供了对结构和功能之间关系的基本理解。本研究证明了 (i) 基质和 PEI 之间对 CO 2 -物理吸附 (φ) 和 CO 2 -化学吸附 (χ) 的竞争,(ii) 滞后 (  )在低温下捕获CO 2归因于CO 2扩散( D )通过PEI膜/塞的动力学( K )阻碍限制了化学吸附,以及(iii)热力学(θ)平衡限制了在高温下的捕获。与 SBA-PEI 材料不同的是,RF 中的第一层 PEI 很容易用于 CO 2捕获,因为该基质不会像 SBA 那样共价结合 PEI。一种简便的方法可以区分物理吸附和化学吸附,并展示前者如何随着 PEI 覆盖而减少。 CO 2捕获滞后虽然很少被介绍或讨论,但强调了普遍接受的“最大捕获”操作温度是基于不完整的实验。通过等温吸附分析,我们将这个最大值的演变与 PEI 的形态分布联系起来。 这一贡献突出了 RF-PEI 的特殊性以及我们的 TGA 协议在理解此类材料的结构/功能关系方面的优势,避免了 SBA 特定协议的典型直接应用。该方法简单,不需要大型设备,并且适用于其他材料。其简便、快速,适合大批量研究,适合对基质性质、孔径、PEI重量等相互作用因素进行综合评价。
)在低温下捕获CO 2归因于CO 2扩散( D )通过PEI膜/塞的动力学( K )阻碍限制了化学吸附,以及(iii)热力学(θ)平衡限制了在高温下的捕获。与 SBA-PEI 材料不同的是,RF 中的第一层 PEI 很容易用于 CO 2捕获,因为该基质不会像 SBA 那样共价结合 PEI。一种简便的方法可以区分物理吸附和化学吸附,并展示前者如何随着 PEI 覆盖而减少。 CO 2捕获滞后虽然很少被介绍或讨论,但强调了普遍接受的“最大捕获”操作温度是基于不完整的实验。通过等温吸附分析,我们将这个最大值的演变与 PEI 的形态分布联系起来。 这一贡献突出了 RF-PEI 的特殊性以及我们的 TGA 协议在理解此类材料的结构/功能关系方面的优势,避免了 SBA 特定协议的典型直接应用。该方法简单,不需要大型设备,并且适用于其他材料。其简便、快速,适合大批量研究,适合对基质性质、孔径、PEI重量等相互作用因素进行综合评价。
 "点击查看英文标题和摘要"
"点击查看英文标题和摘要"
The Structure–Function Relationship of Branched Polyethylenimine Impregnated over Mesoporous Carbon Aerogels: An In-Depth Thermogravimetric Insight
We present a comprehensive thermogravimetric analysis (TGA) of polyethylenimine (PEI)-impregnated resorcinol-formaldehyde (RF) aerogels. While numerous studies focus on PEI-impregnated SBA, RF materials have been less examined, despite their interest and specificities. As most articles on PEI-impregnated porous materials follow typical experimental methods defined for SBA, particularities of RF-PEI materials could remain unheeded. The design of nonisothermal TGA protocols, completed with nitrogen isotherms, based on the systematic filling of the matrix delivers a fundamental understanding of the relationship between the structure and function. This study demonstrates (i) the competition between the matrix and PEI for CO2-physisorption (φ) and CO2-chemisorption (χ), (ii) the hysteresis ( ) of CO2 capture at low temperature attributed to the kinetic (K) hindrance of CO2 diffusion (D) through PEI film/plugs limiting the chemisorption, and (iii) the thermodynamic (θ) equilibrium limiting the capture at high temperature. At variance with SBA-PEI materials, the first layers of PEI in RF are readily available for CO2 capture given that this matrix does not covalently bind PEI as SBA. A facile method allows the discrimination between physi- and chemisorption, exhibiting how the former decreases with PEI coverage. The CO2 capture hysteresis, while seldom introduced or discussed, underlines that the commonly accepted operating temperature of the “maximum capture” is based on an incomplete experiment. Through isotherm adsorption analysis, we correlate the evolution of this maximum to the morphological distribution of PEI. This contribution highlights the specificities of RF-PEI and the advantages of our TGA protocol in understanding the structure/function relationship of this kind of material by avoiding the typical direct applications of SBA-specific protocols. The method is straightforward, does not need large-scale facilities, and is applicable to other materials. Its easiness and rapidness are suited to high-volume studies, befitting for the comprehensive evaluation of interacting factors such as the matrix′s nature, pore size, and PEI weight.
) of CO2 capture at low temperature attributed to the kinetic (K) hindrance of CO2 diffusion (D) through PEI film/plugs limiting the chemisorption, and (iii) the thermodynamic (θ) equilibrium limiting the capture at high temperature. At variance with SBA-PEI materials, the first layers of PEI in RF are readily available for CO2 capture given that this matrix does not covalently bind PEI as SBA. A facile method allows the discrimination between physi- and chemisorption, exhibiting how the former decreases with PEI coverage. The CO2 capture hysteresis, while seldom introduced or discussed, underlines that the commonly accepted operating temperature of the “maximum capture” is based on an incomplete experiment. Through isotherm adsorption analysis, we correlate the evolution of this maximum to the morphological distribution of PEI. This contribution highlights the specificities of RF-PEI and the advantages of our TGA protocol in understanding the structure/function relationship of this kind of material by avoiding the typical direct applications of SBA-specific protocols. The method is straightforward, does not need large-scale facilities, and is applicable to other materials. Its easiness and rapidness are suited to high-volume studies, befitting for the comprehensive evaluation of interacting factors such as the matrix′s nature, pore size, and PEI weight.
更新日期:2023-11-17
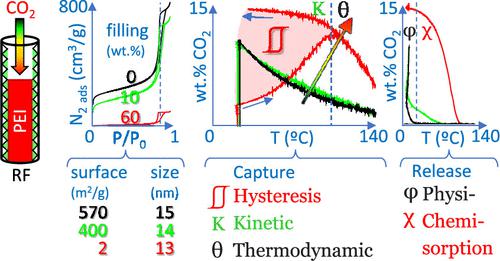
 )在低温下捕获CO 2归因于CO 2扩散( D )通过PEI膜/塞的动力学( K )阻碍限制了化学吸附,以及(iii)热力学(θ)平衡限制了在高温下的捕获。与 SBA-PEI 材料不同的是,RF 中的第一层 PEI 很容易用于 CO 2捕获,因为该基质不会像 SBA 那样共价结合 PEI。一种简便的方法可以区分物理吸附和化学吸附,并展示前者如何随着 PEI 覆盖而减少。 CO 2捕获滞后虽然很少被介绍或讨论,但强调了普遍接受的“最大捕获”操作温度是基于不完整的实验。通过等温吸附分析,我们将这个最大值的演变与 PEI 的形态分布联系起来。 这一贡献突出了 RF-PEI 的特殊性以及我们的 TGA 协议在理解此类材料的结构/功能关系方面的优势,避免了 SBA 特定协议的典型直接应用。该方法简单,不需要大型设备,并且适用于其他材料。其简便、快速,适合大批量研究,适合对基质性质、孔径、PEI重量等相互作用因素进行综合评价。
)在低温下捕获CO 2归因于CO 2扩散( D )通过PEI膜/塞的动力学( K )阻碍限制了化学吸附,以及(iii)热力学(θ)平衡限制了在高温下的捕获。与 SBA-PEI 材料不同的是,RF 中的第一层 PEI 很容易用于 CO 2捕获,因为该基质不会像 SBA 那样共价结合 PEI。一种简便的方法可以区分物理吸附和化学吸附,并展示前者如何随着 PEI 覆盖而减少。 CO 2捕获滞后虽然很少被介绍或讨论,但强调了普遍接受的“最大捕获”操作温度是基于不完整的实验。通过等温吸附分析,我们将这个最大值的演变与 PEI 的形态分布联系起来。 这一贡献突出了 RF-PEI 的特殊性以及我们的 TGA 协议在理解此类材料的结构/功能关系方面的优势,避免了 SBA 特定协议的典型直接应用。该方法简单,不需要大型设备,并且适用于其他材料。其简便、快速,适合大批量研究,适合对基质性质、孔径、PEI重量等相互作用因素进行综合评价。


































 京公网安备 11010802027423号
京公网安备 11010802027423号