Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
调节 DNA 与球形核酸的解离以增强免疫刺激
ACS Nano ( IF 15.8 ) Pub Date : 2023-09-15 , DOI: 10.1021/acsnano.3c04333 Jasper W Dittmar 1 , Michelle H Teplensky 2 , Michael Evangelopoulos 1 , Lei Qin 3 , Bin Zhang 3 , Chad A Mirkin 1, 2
Affiliation
|
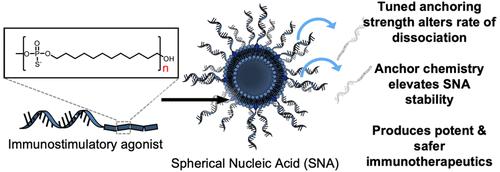
核心的稳定性可以显着影响脂质体药物的治疗效果。虽然球形核酸 (SNA) 结构提高了脂质体稳定性以提高治疗功效,但用于将 DNA 锚定到脂质体核心的化学物质是一个尚未充分研究的设计参数,具有潜在广泛的生物学影响。在此,我们通过系统研究疏水性十二烷锚定基团在将 DNA 链附着到脂质体核心方面的重要性,探讨了 SNA 锚定化学对免疫治疗功能的影响。通过有意调节定义锚定的寡聚物的大小,建立了结构库。这些结构与体外和体内免疫刺激分析相结合,阐明了 DNA 与 SNA 壳的锚定强度和解离对其生物学特性之间的关系和重要性。重要的是,最稳定的十二烷锚定物 (C12) 9优于n = 4-8 和 10 结构,与传统的胆固醇锚定 SNA 相比,免疫刺激增加了四倍。当 OVA1 肽抗原被 (C12) 9 SNA 封装并用作 E.G7-OVA 肿瘤模型中的治疗性疫苗时,50% 的小鼠在最初的肿瘤中存活下来,并且所有这些小鼠都在肿瘤再攻击中存活下来。重要的是,与线性免疫刺激DNA相比,强烈的先天免疫刺激不会引起细胞因子风暴。此外,封装了针对 SARS-CoV-2 的肽的 (C12) 9 SNA 会产生强大的 T 细胞反应; SNA 处理产生的 T 细胞可杀死 >40% 的用相同肽脉冲的靶细胞,并且约 10%。45% 的靶细胞表达整个刺突蛋白。这项工作强调了使用锚定化学来提高 SNA 稳定性的重要性,以在癌症和传染病的背景下实现更有效、更安全的免疫治疗。

"点击查看英文标题和摘要"













































 京公网安备 11010802027423号
京公网安备 11010802027423号