European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2023-07-19 , DOI: 10.1016/j.ejmech.2023.115637 Kyle D Farrell 1 , Yamin Gao 2 , Deborah A Hughes 3 , Robin Henches 3 , Zhengchao Tu 4 , Michael V Perkins 1 , Tianyu Zhang 2 , Craig L Francis 3
|
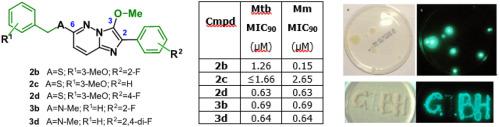
A series of 3-methoxy-2-phenylimidazo[1,2-b]pyridazine derivatives which were highly active against autoluminescent Mycobacterium tuberculosis (Mtb) and Mycobacterium marinum (Mm) in an in vitro assay were identified. SAR analysis showed that the most active compounds, which included a phenyl group bearing fluoro substituent(s) at C2, a methoxy function at C3, and a benzyl-heteroatom moiety at C6, exhibited in vitro MIC90 values generally around 0.63–1.26 μM against Mtb and Mm. However, these compounds were inactive against Mtb in vivo (mice), and investigations revealed very short metabolic half-lives (<10 min) when incubated with mouse liver microsomes. Multiple observations of side products produced from oxidative cleavage of the imidazole moiety during the chemical synthesis work suggested that this is a likely metabolic pathway leading to the lack of observed activity in vivo.
中文翻译:

3-甲氧基-2-苯基咪唑并[1,2-b]哒嗪对结核分枝杆菌和海分枝杆菌具有高度活性
在体外测定中鉴定出一系列 3-甲氧基-2-苯基咪唑并[1,2- b ]哒嗪衍生物,它们对自发光结核分枝杆菌(Mtb) 和海分枝杆菌(Mm) 具有高度活性。SAR 分析显示,最活跃的化合物(包括 C2 处带有氟取代基的苯基、C3 处的甲氧基官能团和 C6 处的苄基杂原子部分)表现出的体外 MIC 90 值通常约为0.63–1.26 μM对抗 Mtb 和 Mm。然而,这些化合物在体内(小鼠)对 Mtb 没有活性,并且研究表明,当与小鼠肝微粒体一起孵育时,代谢半衰期非常短(<10 分钟)。对化学合成过程中咪唑部分氧化裂解产生的副产物的多次观察表明,这可能是导致体内缺乏观察到的活性的代谢途径。

































 京公网安备 11010802027423号
京公网安备 11010802027423号