Phytomedicine ( IF 6.7 ) Pub Date : 2023-06-02 , DOI: 10.1016/j.phymed.2023.154912 Zi-Qi Dai 1 , Feng Gao 1 , Zi-Jie Zhang 1 , Ming-Jun Lu 1 , Yu-Jin Luo 1 , Tong Zhang 1 , Bing-Xian Shang 1 , Yu-Hao Gu 1 , Qi Zeng 1 , Shan Gao 1 , Zhuo-Qian Guo 1 , Bing Xu 1 , Hai-Min Lei 1
|
背景
基于肿瘤细胞糖酵解和能量代谢的治疗方法是治疗癌症的新的有前途的策略。目前,抑制糖酵解中关键限速酶丙酮酸激酶M2的研究已被证实是一种有效的癌症治疗方法。Alkannin 是一种有效的丙酮酸激酶 M2 抑制剂。然而,其非选择性细胞毒性影响了其后续的临床应用。因此,需要对其进行结构修饰以开发具有高选择性的新型衍生物。
目的
我们的研究旨在通过结构修饰改善 alkannin 的毒性,并阐明其优越衍生物 23 在肺癌治疗中的作用机制。
方法
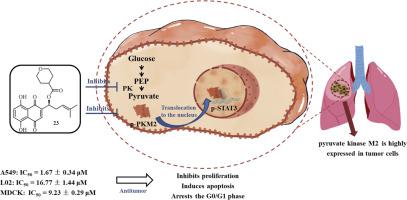
根据配置原理,将不同的氨基酸和含氧杂环引入到alkannin侧链的羟基中。我们通过MTT 测定检测了所有衍生物对三种肿瘤细胞(HepG2、A549 和 HCT116)和两种正常细胞(L02 和 MDCK)的细胞活力。此外,分别通过姬姆萨染色和DAPI染色观察到衍生物23对A549细胞形态的影响。进行流式细胞术以评估衍生物 23 对细胞凋亡和细胞周期停滞的影响。为了进一步评估衍生物 23 对糖酵解中丙酮酸激酶 M2 的影响,进行了酶活性最后,体内利用Lewis小鼠肺癌异种移植模型评价衍生物23的抗肿瘤活性和安全性。
结果
设计并合成了 23 种新型 alkannin 衍生物,以提高细胞毒性选择性。在这些衍生物中,衍生物 23 在癌细胞和正常细胞之间表现出最高的细胞毒性选择性。衍生物 23 对 A549 细胞的抗增殖活性 (IC 50 = 1.67 ± 0.34 μM) 比 L02 细胞 (IC 50 = 16.77 ± 1.44 μM) 高 10 倍,比 MDCK 细胞 (IC 50 = 9.23 ) 高 5 倍± 0.29 μM)分别。随后,荧光染色流式细胞仪分析表明,衍生物23能够诱导A549细胞凋亡,使细胞周期停滞在G0/G1期。此外,机理研究表明衍生物 23 是丙酮酸激酶的抑制剂;它可以通过抑制 PKM2/STAT3 信号通路磷酸化的激活来调节糖酵解。此外,体内研究表明衍生物 23 显着抑制异种移植肿瘤的生长。
结论
在这项研究中,alkannin 选择性被报道在结构修饰后显着提高,衍生物 23 首次显示能够在体外通过 PKM2/STAT3 磷酸化信号通路抑制肺癌生长,表明衍生物 23 在治疗肺癌方面的潜在价值肺癌。

"点击查看英文标题和摘要"































 京公网安备 11010802027423号
京公网安备 11010802027423号