Pesticide Biochemistry and Physiology ( IF 4.2 ) Pub Date : 2023-05-26 , DOI: 10.1016/j.pestbp.2023.105480 Shujie Ma 1 , Hualong Yu 1 , Mingyu Wang 1 , Tingru Cui 2 , Yujing Zhao 1 , Xinxin Zhang 1 , Caixia Wang 1 , Mengmeng Li 1 , Lihui Zhang 1 , Jingao Dong 1
|
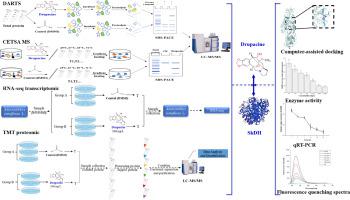
Natural products are one of the important sources for the creation of new pesticides. Drupacine ((1R,11S,12S,13R,15S)-13-methoxy-5,7,21-trioxa-19-azahexacyclo[11.7.1.02,10.04,8.011,15.015,19]henicosa-2,4(8),9-trien-12-ol), isolated from Cephalotaxus sinensis (Chinese plum-yew), is a potent herbicidal compound containing an oxo-bridged oxygen bond structure. However, its molecular target still remains unknown. In this study, the targets of drupacine in Amaranthus retroflexus were identified by combining drug affinity responsive target stability (DARTS), cellular thermal shift assay coupled with mass spectrometry (CETSA MS), RNA-seq transcriptomic, and TMT proteomic analyses. Fifty-one and sixty-eight main binding proteins were identified by DARTS and CETSA MS, respectively, including nine co-existing binding proteins. In drupacine-treated A. retroflexus seedlings we identified 1389 up-regulated genes and 442 down-regulated genes, 34 up-regulated proteins, and 194 down-regulated proteins, respectively. Combining the symptoms and the biochemical profiles, Profilin, Shikimate dehydrogenase (SkDH), and Zeta-carotene desaturase were predicted to be the drupacine potential target proteins. At the same time, drupacine was found to bind SkDH stronger by molecular docking, and its inhibition on ArSkDH increased with the treatment concentration increase. Our results suggest that the molecular target of drupacine is SkDH, a new herbicide target, which lay a foundation for the rational design of herbicides based on new targets from natural products and enrich the target resources for developing green herbicides.































 京公网安备 11010802027423号
京公网安备 11010802027423号