当前位置:
X-MOL 学术
›
Green Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Electrochemical intramolecular N(sp2)–H/N(sp3)–H coupling for the synthesis of 1H-indazoles
Green Chemistry ( IF 9.3 ) Pub Date : 2023-05-02 , DOI: 10.1039/d3gc00380a
Qiang Zhong 1 , Pei-Long Wang 1, 2 , Hui Gao 1, 3 , Fang Ma 1 , Youqing Yang 1 , Hongji Li 1
Green Chemistry ( IF 9.3 ) Pub Date : 2023-05-02 , DOI: 10.1039/d3gc00380a
Qiang Zhong 1 , Pei-Long Wang 1, 2 , Hui Gao 1, 3 , Fang Ma 1 , Youqing Yang 1 , Hongji Li 1
Affiliation
|
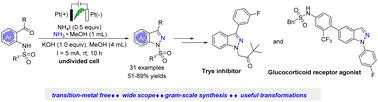
The electrochemical synthesis of 1H-indazoles via an intramolecular N(sp2)–H/N(sp3)–H coupling reaction at room temperature is first described. The transition metal-free electrochemical cyclization features the use of commercially available NH3 as the nitrogen source and a broad substrate scope, thus affording a wide range of substituted 1H-indazoles in moderate to good yields. Both the control experiments and mechanistic studies support the proposed reaction mechanism for this electrochemical cyclization.
中文翻译:

电化学分子内 N(sp2)–H/N(sp3)–H 偶联合成 1H-吲唑
首先描述了在室温下通过分子内 N(sp 2 )–H/N(sp 3 )–H 偶联反应电化学合成 1 H-吲唑。无过渡金属电化学环化的特点是使用市售的 NH 3作为氮源和广泛的底物范围,从而以中等到良好的产率提供范围广泛的取代 1 H-吲唑。控制实验和机理研究都支持所提出的这种电化学环化反应机理。
更新日期:2023-05-02
中文翻译:

电化学分子内 N(sp2)–H/N(sp3)–H 偶联合成 1H-吲唑
首先描述了在室温下通过分子内 N(sp 2 )–H/N(sp 3 )–H 偶联反应电化学合成 1 H-吲唑。无过渡金属电化学环化的特点是使用市售的 NH 3作为氮源和广泛的底物范围,从而以中等到良好的产率提供范围广泛的取代 1 H-吲唑。控制实验和机理研究都支持所提出的这种电化学环化反应机理。

































 京公网安备 11010802027423号
京公网安备 11010802027423号