当前位置:
X-MOL 学术
›
ACS Chem. Biol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Biosynthesis-Guided Discovery and Engineering of α-Pyrone Natural Products from Type I Polyketide Synthases
ACS Chemical Biology ( IF 3.5 ) Pub Date : 2023-04-19 , DOI: 10.1021/acschembio.3c00081 Dongqi Yi 1 , Vinayak Agarwal 1, 2
ACS Chemical Biology ( IF 3.5 ) Pub Date : 2023-04-19 , DOI: 10.1021/acschembio.3c00081 Dongqi Yi 1 , Vinayak Agarwal 1, 2
Affiliation
|
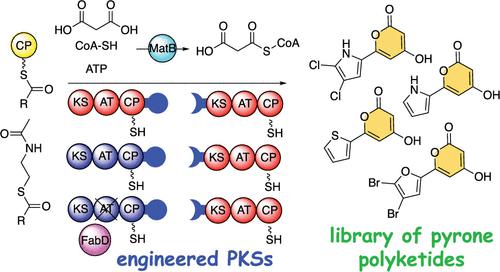
Natural products containing the α-pyrone moiety are produced by polyketide synthases (PKSs) in bacteria, fungi, and plants. The conserved biosynthetic logic for the production of the α-pyrone moiety involves the cyclization of a triketide intermediate which also off-loads the polyketide from the activating thioester. In this study, we show that truncating a tetraketide natural product producing PKS assembly line allows for a thioesterase-independent off-loading of an α-pyrone polyketide natural product, one which we find to be natively present in the extracts of the bacterium that otherwise furnishes the tetraketide natural product. By engineering the truncated PKS in vitro, we demonstrate that a ketosynthase (KS) domain with relaxed substrate selectivity when coupled with in trans acylation of polyketide extender units can expand the chemical space of α-pyrone polyketide natural products. Findings from this study point toward heterologous intermolecular protein–protein interactions being detrimental to the efficiency of engineered PKS assembly lines.
中文翻译:

I 型聚酮合成酶的 α-吡喃酮天然产物的生物合成引导发现和工程
含有 α-吡喃酮部分的天然产物由细菌、真菌和植物中的聚酮合酶 (PKS) 产生。用于生产 α-吡喃酮部分的保守生物合成逻辑涉及三酮中间体的环化,该中间体还从活化硫酯中卸载聚酮化合物。在这项研究中,我们表明,截断生产四酮化合物天然产物的 PKS 装配线可以不依赖于硫酯酶的 α-吡喃酮聚酮化合物天然产物的卸载,我们发现它天然存在于细菌的提取物中,否则提供四酮天然产物。通过在体外设计截短的 PKS,我们证明,当与聚酮化合物扩展单元的反式酰基化相结合时,具有松弛底物选择性的酮合成酶 (KS) 结构域可以扩展 α-吡喃酮聚酮化合物天然产物的化学空间。这项研究的结果表明,异源分子间蛋白质-蛋白质相互作用不利于工程化 PKS 装配线的效率。
更新日期:2023-04-19
中文翻译:

I 型聚酮合成酶的 α-吡喃酮天然产物的生物合成引导发现和工程
含有 α-吡喃酮部分的天然产物由细菌、真菌和植物中的聚酮合酶 (PKS) 产生。用于生产 α-吡喃酮部分的保守生物合成逻辑涉及三酮中间体的环化,该中间体还从活化硫酯中卸载聚酮化合物。在这项研究中,我们表明,截断生产四酮化合物天然产物的 PKS 装配线可以不依赖于硫酯酶的 α-吡喃酮聚酮化合物天然产物的卸载,我们发现它天然存在于细菌的提取物中,否则提供四酮天然产物。通过在体外设计截短的 PKS,我们证明,当与聚酮化合物扩展单元的反式酰基化相结合时,具有松弛底物选择性的酮合成酶 (KS) 结构域可以扩展 α-吡喃酮聚酮化合物天然产物的化学空间。这项研究的结果表明,异源分子间蛋白质-蛋白质相互作用不利于工程化 PKS 装配线的效率。

































 京公网安备 11010802027423号
京公网安备 11010802027423号