当前位置:
X-MOL 学术
›
J. Med. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Discovery and Optimization of 1-Phenoxy-2-aminoindanes as Potent, Selective, and Orally Bioavailable Inhibitors of the Na+/H+ Exchanger Type 3 (NHE3)
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2016-09-22 00:00:00 , DOI: 10.1021/acs.jmedchem.6b00624
Nils Rackelmann 1 , Hans Matter 1 , Heinrich Englert 1 , Markus Follmann 1 , Thomas Maier 1 , John Weston 1 , Petra Arndt 1 , Winfried Heyse 1 , Katharina Mertsch 1 , Klaus Wirth 1 , Laurent Bialy 1
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2016-09-22 00:00:00 , DOI: 10.1021/acs.jmedchem.6b00624
Nils Rackelmann 1 , Hans Matter 1 , Heinrich Englert 1 , Markus Follmann 1 , Thomas Maier 1 , John Weston 1 , Petra Arndt 1 , Winfried Heyse 1 , Katharina Mertsch 1 , Klaus Wirth 1 , Laurent Bialy 1
Affiliation
|
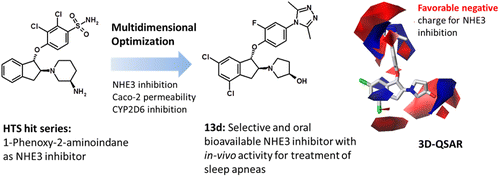
The design, synthesis, and structure–activity relationship of 1-phenoxy-2-aminoindanes as inhibitors of the Na+/H+ exchanger type 3 (NHE3) are described based on a hit from high-throughput screening (HTS). The chemical optimization resulted in the discovery of potent, selective, and orally bioavailable NHE3 inhibitors with 13d as best compound, showing high in vitro permeability and lacking CYP2D6 inhibition as main optimization parameters. Aligning 1-phenoxy-2-aminoindanes onto the X-ray structure of 13d then provided 3D-QSAR models for NHE3 inhibition capturing guidelines for optimization. These models showed good correlation coefficients and allowed for activity estimation. In silico ADMET models for Caco-2 permeability and CYP2D6 inhibition were also successfully applied for this series. Moreover, docking into the CYP2D6 X-ray structure provided a reliable alignment for 3D-QSAR models. Finally 13d, renamed as SAR197, was characterized in vitro and by in vivo pharmacokinetic (PK) and pharmacological studies to unveil its potential for reduction of obstructive sleep apneas.
中文翻译:

发现和优化1-苯氧基-2-氨基茚满作为Na + / H + 3型交换子(NHE3)的有效,选择性和口服生物利用抑制剂
基于高通量筛选(HTS)的命中,描述了作为3 + Na + / H +交换剂(NHE3)抑制剂的1-苯氧基-2-氨基茚满的设计,合成及其构效关系。化学最优化导致发现了以13d为最佳化合物的有效,选择性和口服生物利用的NHE3抑制剂,显示出较高的体外通透性和缺乏CYP2D6抑制作用作为主要优化参数。将1-苯氧基-2-氨基茚满对准13d的X射线结构然后为NHE3抑制捕获指南提供了3D-QSAR模型以进行优化。这些模型显示出良好的相关系数,并可以进行活性估算。在计算机中针对Caco-2渗透性和CYP2D6抑制作用的ADMET模型也已成功应用于该系列。此外,对接CYP2D6 X射线结构为3D-QSAR模型提供了可靠的对齐方式。最终,重命名为SAR197的13d在体外和体内药代动力学(PK)和药理研究中进行了表征,以揭示其减少阻塞性睡眠呼吸暂停的潜力。
更新日期:2016-09-22
中文翻译:

发现和优化1-苯氧基-2-氨基茚满作为Na + / H + 3型交换子(NHE3)的有效,选择性和口服生物利用抑制剂
基于高通量筛选(HTS)的命中,描述了作为3 + Na + / H +交换剂(NHE3)抑制剂的1-苯氧基-2-氨基茚满的设计,合成及其构效关系。化学最优化导致发现了以13d为最佳化合物的有效,选择性和口服生物利用的NHE3抑制剂,显示出较高的体外通透性和缺乏CYP2D6抑制作用作为主要优化参数。将1-苯氧基-2-氨基茚满对准13d的X射线结构然后为NHE3抑制捕获指南提供了3D-QSAR模型以进行优化。这些模型显示出良好的相关系数,并可以进行活性估算。在计算机中针对Caco-2渗透性和CYP2D6抑制作用的ADMET模型也已成功应用于该系列。此外,对接CYP2D6 X射线结构为3D-QSAR模型提供了可靠的对齐方式。最终,重命名为SAR197的13d在体外和体内药代动力学(PK)和药理研究中进行了表征,以揭示其减少阻塞性睡眠呼吸暂停的潜力。







































 京公网安备 11010802027423号
京公网安备 11010802027423号