Separation and Purification Technology ( IF 8.1 ) Pub Date : 2023-01-24 , DOI: 10.1016/j.seppur.2023.123272 Yixuan Fan , Jie Zhang , Lixia Yang , Mingxia Lu , Tiantian Ying , Banghong Deng , Weili Dai , Xubiao Luo , Jianping Zou , Shenglian Luo
|
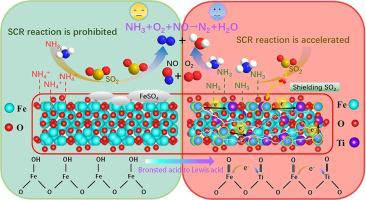
An ideal catalyst that can be used for the selective catalytic reduction (SCR) of NO at low temperature should have a high activity and ability to resist SO2. Herein, Ti-doped Fe2O3 (Ti-Fe2O3) nanoparticles that extraordinarily convert NO (T90, 200 °C) and highly resist SO2 meet this requirement. In the presence of 100 ppm SO2, the optimal Ti-Fe2O3 can convert 90% NO at 220 °C for 24 h. With the addition of Ti4+, the electrons around Fe3+ migrate toward Ti4+, strengthening the electron-deficient tendency of Fe3+. Therefore, electron-donating NH3 is preferentially adsorbed by Fe3+, endowing Ti-Fe2O3 with the ability to resist SO2 and exceptionally tolerate SO2 at low temperature. Besides, based on the strength of the embedded Ti4+, a large number of Brønsted acid sites are converted to Lewis acid sites to speed the reaction rate of SCR below 210 °C. High anti-SO2 capacity and adequate Lewis acid sites maximize the efficiency of Ti-Fe2O3 in reducing NO.













































 京公网安备 11010802027423号
京公网安备 11010802027423号