当前位置:
X-MOL 学术
›
Green Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Sodium dithionite mediated one-pot, tandem chemoselective reduction/cyclization for the synthesis of pyrrole fused N-heterocycles
Green Chemistry ( IF 9.3 ) Pub Date : 2022-12-02 , DOI: 10.1039/d2gc03749a Joydev K. Laha 1 , Surabhi Panday 1 , Pankaj Gupta 1 , Shiv Raj Seth 1
Green Chemistry ( IF 9.3 ) Pub Date : 2022-12-02 , DOI: 10.1039/d2gc03749a Joydev K. Laha 1 , Surabhi Panday 1 , Pankaj Gupta 1 , Shiv Raj Seth 1
Affiliation
|
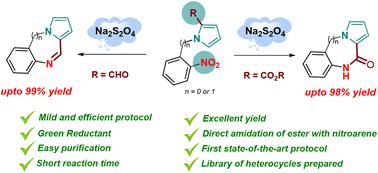
A chemoselective reduction of a nitro group in the presence of an aldehyde or ester group integrated with another synthetic transformation leading to the expedient synthesis of important heterocycles is the subject of this current investigation. The chemoselective reductive cyclization of N-(2-nitrophenyl)pyrrole-2-carboxaldehydes accompanied exclusively by an industrial reagent sodium dithionite (Na2S2O4) yielding diversely substituted pyrrole fused N-heterocycles has been developed for the first time. The amino group generated in situ via chemoselective reduction of the nitro group undergoes condensation with the aldehyde group to form quinoxalines, or undergoes reaction with the ester group to form quinoxalones. The protocol features an efficient one-pot, tandem reductive cyclization performed at room temperature, very short reaction time (1 h), no aqueous work up, purification by crystallization, isolated yields generally >90%, appreciable functional group tolerance, and wide substrate scope. The scalability of the developed protocol is further demonstrated by a gram-scale synthesis.
中文翻译:

连二亚硫酸钠介导的一锅串联化学选择性还原/环化用于合成吡咯稠合 N-杂环
在醛基或酯基存在的情况下,硝基的化学选择性还原与另一种合成转化相结合,从而方便地合成重要的杂环,是当前研究的主题。N -(2-硝基苯基)吡咯-2-甲醛的化学选择性还原环化仅伴随工业试剂连二亚硫酸钠 (Na 2 S 2 O 4 ) 产生不同取代的吡咯稠合N -杂环已首次开发。原位生成的氨基通过化学选择性还原硝基与醛基缩合形成喹喔啉,或与酯基反应形成喹喔啉酮。该协议具有在室温下进行的高效单锅串联还原环化、非常短的反应时间(1 小时)、无水后处理、结晶纯化、分离产率通常 >90%、可观的官能团耐受性和广泛的底物范围。克级合成进一步证明了所开发协议的可扩展性。
更新日期:2022-12-02
中文翻译:

连二亚硫酸钠介导的一锅串联化学选择性还原/环化用于合成吡咯稠合 N-杂环
在醛基或酯基存在的情况下,硝基的化学选择性还原与另一种合成转化相结合,从而方便地合成重要的杂环,是当前研究的主题。N -(2-硝基苯基)吡咯-2-甲醛的化学选择性还原环化仅伴随工业试剂连二亚硫酸钠 (Na 2 S 2 O 4 ) 产生不同取代的吡咯稠合N -杂环已首次开发。原位生成的氨基通过化学选择性还原硝基与醛基缩合形成喹喔啉,或与酯基反应形成喹喔啉酮。该协议具有在室温下进行的高效单锅串联还原环化、非常短的反应时间(1 小时)、无水后处理、结晶纯化、分离产率通常 >90%、可观的官能团耐受性和广泛的底物范围。克级合成进一步证明了所开发协议的可扩展性。













































 京公网安备 11010802027423号
京公网安备 11010802027423号