Journal of Molecular Structure ( IF 4.0 ) Pub Date : 2022-06-20 , DOI: 10.1016/j.molstruc.2022.133552 Rajendran Satheeshkumar , Kolandaivel Prabha , Kailasam Natesan Vennila , Koray Sayin , Elif Güney , Werner Kaminsky , Roberto Acevedo
|
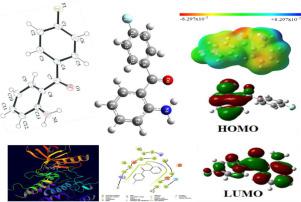
In this work, synthesis, and crystal structure of molecule 2-amino-4′-fluorobenzophenone (FAB) is confirmed by using FT-IR, FT-Raman, 1H and 13C NMR chemical shifts, compared with calculated parameters using B3LYP/ 6-311+G(d) basis sets in water were found in good agreement. The optimized geometry of the molecule (FAB) was compared to the experimental XRD values. DFT calculations of the molecular electrostatic potential (MEP), frontier molecular orbitals (FMO), Hirshfeld surface analysis, Mulliken charges recognize the chemically active sites of this molecule responsible for its chemical reactivity. In silico molecular docking analyses of molecule (FAB) have been done with vascular endothelial growth factor receptor 2 (VEGFR2) kinase inhibitors. Further, the bioavailability of molecule (FAB) was investigated by ADME and p450 analyses.
中文翻译:

光谱(FT-IR、NMR、单晶 XRD)和 DFT 研究,包括 2-氨基-4'-氟二苯甲酮 (FAB) 的 FMO、Mulliken 电荷和 Hirshfeld 表面分析、分子对接和 ADME 分析
在这项工作中,分子 2-氨基-4'-氟二苯甲酮 (FAB) 的合成和晶体结构通过使用 FT-IR、FT-拉曼、1 H 和13 C NMR 化学位移与使用 B3LYP/发现水中的 6-311+G(d) 基组非常一致。将优化的分子几何形状 (FAB) 与实验 XRD 值进行比较。分子静电势 (MEP)、前沿分子轨道 (FMO) 的 DFT 计算、Hirshfeld 表面分析、Mulliken 电荷识别负责其化学反应性的该分子的化学活性位点。计算机已经用血管内皮生长因子受体 2 (VEGFR2) 激酶抑制剂对分子 (FAB) 进行了分子对接分析。此外,通过 ADME 和 p450 分析研究了分子 (FAB) 的生物利用度。










































 京公网安备 11010802027423号
京公网安备 11010802027423号