当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Localized Antiaromaticity Hotspot Drives Reductive Dehydrogenative Cyclizations in Bis- and Mono-Helicenes
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-06-02 , DOI: 10.1021/jacs.2c03681
Zheng Zhou 1, 2 , Dominic T Egger 3 , Chaowei Hu 4 , Matthew Pennachio 1 , Zheng Wei 1 , Rahul K Kawade 4 , Ökten Üngör 4 , Renana Gershoni-Poranne 3, 5 , Marina A Petrukhina 1 , Igor V Alabugin 4
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-06-02 , DOI: 10.1021/jacs.2c03681
Zheng Zhou 1, 2 , Dominic T Egger 3 , Chaowei Hu 4 , Matthew Pennachio 1 , Zheng Wei 1 , Rahul K Kawade 4 , Ökten Üngör 4 , Renana Gershoni-Poranne 3, 5 , Marina A Petrukhina 1 , Igor V Alabugin 4
Affiliation
|
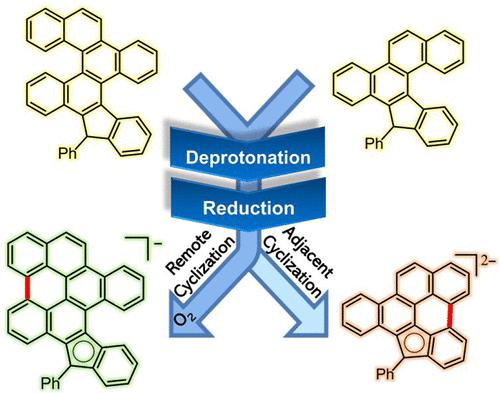
We describe reductive dehydrogenative cyclizations that form hepta-, nona-, and decacyclic anionic graphene subunits from mono- and bis-helicenes with an embedded five-membered ring. The reaction of bis-helicenes can either proceed to the full double annulation or be interrupted by addition of molecular oxygen at an intermediate stage. The regioselectivity of the interrupted cyclization cascade for bis-helicenes confirms that relief of antiaromaticity is a dominant force for these facile ring closures. Computational analysis reveals the unique role of the preexisting negatively charged cyclopentadienyl moiety in directing the second negative charge at a specific remote location and, thus, creating a localized antiaromatic region. This region is the hotspot that promotes the initial cyclization. Computational studies, including MO analysis, molecular electrostatic potential maps, and NICS(1.7)ZZ calculations, evaluate the interplay of the various effects including charge delocalization, helicene strain release, and antiaromaticity. The role of antiaromaticity relief is further supported by efficient reductive closure of the less strained monohelicenes where the relief of antiaromaticity promotes the cyclization even when the strain is substantially reduced. The latter finding significantly expands the scope of this reductive alternative to the Scholl ring closure.
中文翻译:

局部反芳烃热点驱动双螺旋和单螺旋中的还原脱氢环化
我们描述了从具有嵌入五元环的单螺旋和双螺旋烯形成七环、九环和十环阴离子石墨烯亚基的还原脱氢环化。双螺旋的反应可以进行到完全双环化,也可以通过在中间阶段添加分子氧来中断。双螺旋的间断环化级联的区域选择性证实了抗芳香性的减轻是这些易闭环的主导力量。计算分析揭示了预先存在的带负电荷的环戊二烯基部分在将第二个负电荷引导到特定的远程位置并因此产生局部反芳烃区域的独特作用。该区域是促进初始环化的热点。计算研究,包括 MO 分析,ZZ计算,评估各种效应的相互作用,包括电荷离域、螺旋烯应变释放和抗芳香性。抗芳香性消除的作用进一步得到了张力较小的单螺旋烯的有效还原闭合的支持,其中抗芳香性的消除促进了环化,即使在应变显着降低时也是如此。后一个发现显着扩展了 Scholl 环闭合的这种还原替代方案的范围。
更新日期:2022-06-02
中文翻译:

局部反芳烃热点驱动双螺旋和单螺旋中的还原脱氢环化
我们描述了从具有嵌入五元环的单螺旋和双螺旋烯形成七环、九环和十环阴离子石墨烯亚基的还原脱氢环化。双螺旋的反应可以进行到完全双环化,也可以通过在中间阶段添加分子氧来中断。双螺旋的间断环化级联的区域选择性证实了抗芳香性的减轻是这些易闭环的主导力量。计算分析揭示了预先存在的带负电荷的环戊二烯基部分在将第二个负电荷引导到特定的远程位置并因此产生局部反芳烃区域的独特作用。该区域是促进初始环化的热点。计算研究,包括 MO 分析,ZZ计算,评估各种效应的相互作用,包括电荷离域、螺旋烯应变释放和抗芳香性。抗芳香性消除的作用进一步得到了张力较小的单螺旋烯的有效还原闭合的支持,其中抗芳香性的消除促进了环化,即使在应变显着降低时也是如此。后一个发现显着扩展了 Scholl 环闭合的这种还原替代方案的范围。


































 京公网安备 11010802027423号
京公网安备 11010802027423号