Journal of Organometallic Chemistry ( IF 2.1 ) Pub Date : 2022-05-30 , DOI: 10.1016/j.jorganchem.2022.122408 Yuvaraja Dibdalli , José Gaete , Cristian Valdebenito , Juan Luis Arroyo , Iván Martínez , Gabriel Abarca , Cesar Morales-Verdejo
|
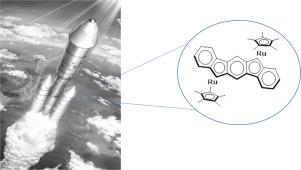
We reported the synthesis of a novel bimetallic compound with the formula [(Cp*Ru)2-IF] derived from 6,12-dihydroindeno[1,2-b]fluorene ligand (IFH2). This compound was characterized by 1H and 13C NMR, elemental analysis, FT-IR, and UV-vis.
The bimetallic compound was tested as a burning rate catalyst on the thermal decomposition of ammonium perchlorate. It was assessed by differential scanning calorimetry technique (DSC) analysis to understand its catalytic behavior further. [(Cp*Ru)2-IF] shows a decrease in the decomposition temperature of AP to 381°C, increasing the energy release to 1271 J•g−1. In addition, this compound leads to the lowest activation energy (20.7 kJ•mol−1), suggesting a suitable and competitive alternative to be used as a modifier for composite solid propellants.
中文翻译:

6,12-二氢茚并[1,2-b]芴配体衍生的双金属钌化合物作为固体火箭发动机推进剂的燃速催化剂
我们报道了一种由 6,12-二氢茚并[1,2-b]芴配体 (IFH 2 ) 衍生的分子式 [(Cp*Ru) 2 -IF]的新型双金属化合物的合成。该化合物通过1 H 和13 C NMR、元素分析、FT-IR 和 UV-vis 进行了表征。
双金属化合物作为燃烧速率催化剂对高氯酸铵的热分解进行了测试。通过差示扫描量热技术 (DSC) 分析对其进行评估,以进一步了解其催化行为。[(Cp*Ru) 2 -IF]表明AP的分解温度降低至381°C,能量释放增加至1271 J•g -1。此外,该化合物的活化能最低(20.7 kJ•mol -1),表明它是一种合适且具有竞争力的替代品,可用作复合固体推进剂的改性剂。






































 京公网安备 11010802027423号
京公网安备 11010802027423号