当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Site-Specific and Degree-Controlled Alkyl Deuteration via Cu-Catalyzed Redox-Neutral Deacylation
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-05-25 , DOI: 10.1021/jacs.2c04382 Xukai Zhou 1 , Tingting Yu 1 , Guangbin Dong 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-05-25 , DOI: 10.1021/jacs.2c04382 Xukai Zhou 1 , Tingting Yu 1 , Guangbin Dong 1
Affiliation
|
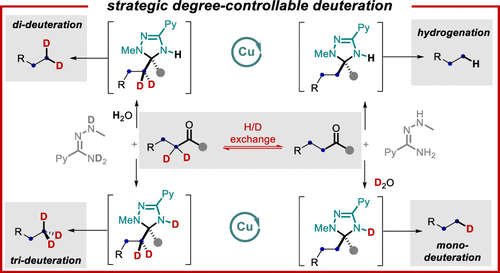
Deuterated organic compounds have become increasingly important in many areas; however, it remains challenging to install deuterium site-selectively to unactivated aliphatic positions with control of the degree of deuteration. Here, we report a Cu-catalyzed degree-controlled deacylative deuteration of diverse alkyl groups with the methylketone (acetyl) moiety as a traceless activating group. The use of N-methylpicolino-hydrazonamide (MPHA) promotes efficient aromatization-driven C–C cleavage. Mono-, di-, and trideuteration at specific sites can be selectively achieved. The reaction is redox-neutral with broad functional group tolerance. The utility of this method has been demonstrated in forming a complete set of deuterated ethyl groups, merging with the Diels–Alder reaction, a net devinylative deuteration, and the synthesis of the d2-analogue of Austedo.
中文翻译:

通过 Cu 催化的氧化还原中性脱酰作用进行位点特异性和程度可控的烷基氘化
氘代有机化合物在许多领域变得越来越重要;然而,通过控制氘化程度将氘选择性地安装到未激活的脂肪族位置仍然具有挑战性。在这里,我们报告了铜催化的不同烷基的度控制脱酰氘化,甲基酮(乙酰基)部分作为无痕活化基团。的使用-methylpicolino-hydrazonamide (MPHA) 促进有效的芳构化驱动的 C-C 裂解。可以选择性地实现特定站点的单、双和三氘化。该反应是氧化还原中性的,具有广泛的官能团耐受性。该方法的实用性已在形成一套完整的氘代乙基、与 Diels-Alder 反应合并、净脱乙烯基氘化以及合成 d 2 -Austedo 类似物方面得到证明。
更新日期:2022-05-25
中文翻译:

通过 Cu 催化的氧化还原中性脱酰作用进行位点特异性和程度可控的烷基氘化
氘代有机化合物在许多领域变得越来越重要;然而,通过控制氘化程度将氘选择性地安装到未激活的脂肪族位置仍然具有挑战性。在这里,我们报告了铜催化的不同烷基的度控制脱酰氘化,甲基酮(乙酰基)部分作为无痕活化基团。的使用-methylpicolino-hydrazonamide (MPHA) 促进有效的芳构化驱动的 C-C 裂解。可以选择性地实现特定站点的单、双和三氘化。该反应是氧化还原中性的,具有广泛的官能团耐受性。该方法的实用性已在形成一套完整的氘代乙基、与 Diels-Alder 反应合并、净脱乙烯基氘化以及合成 d 2 -Austedo 类似物方面得到证明。































 京公网安备 11010802027423号
京公网安备 11010802027423号