当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
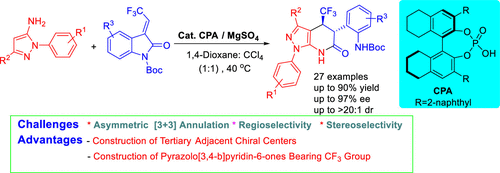
Asymmetric [3 + 3] Annulation to Construct Trifluoromethylated Pyrazolo[3,4-b]pyridin-6-ones via Chiral Phosphoric Acid and MgSO4 Synergistic Catalysis
Organic Letters ( IF 4.9 ) Pub Date : 2022-05-25 , DOI: 10.1021/acs.orglett.2c01513 Alemayehu Gashaw Woldegiorgis 1 , Zhao Han 1 , Xufeng Lin 1
Organic Letters ( IF 4.9 ) Pub Date : 2022-05-25 , DOI: 10.1021/acs.orglett.2c01513 Alemayehu Gashaw Woldegiorgis 1 , Zhao Han 1 , Xufeng Lin 1
Affiliation
|
We developed a novel asymmetric Friedel–Crafts alkylation/transamidation tandem reaction for the enantio- and diastereoselective synthesis of pyrazolo[3,4-b]pyridin-6-ones bearing a −CF3 unit via synergistic chiral phosphoric acid and MgSO4 catalysis. This [3 + 3] annulation protocol allows the formation of trifluoromethylated pyrazolo[3,4-b]pyridin-6-ones with two adjacent tertiary stereocenters in moderate to high yields (up to 90%), enantioselectivities (up to 97% ee), and diastereoselectivities (up to >20:1 dr).
中文翻译:

手性磷酸和MgSO4协同催化不对称[3+3]环化构建三氟甲基吡唑并[3,4-b]吡啶-6-酮
我们开发了一种新颖的不对称 Friedel-Crafts 烷基化/转酰胺基串联反应,用于通过协同手性磷酸和 MgSO 4催化合成带有 -CF 3单元的吡唑并[3,4- b ]吡啶-6-酮的对映选择性和非对映选择性。这种 [3 + 3] 环化方案允许以中等至高产率(高达 90%)、对映选择性(高达 97% ee ) 和非对映选择性(高达 >20:1 dr)。
更新日期:2022-05-25
中文翻译:

手性磷酸和MgSO4协同催化不对称[3+3]环化构建三氟甲基吡唑并[3,4-b]吡啶-6-酮
我们开发了一种新颖的不对称 Friedel-Crafts 烷基化/转酰胺基串联反应,用于通过协同手性磷酸和 MgSO 4催化合成带有 -CF 3单元的吡唑并[3,4- b ]吡啶-6-酮的对映选择性和非对映选择性。这种 [3 + 3] 环化方案允许以中等至高产率(高达 90%)、对映选择性(高达 97% ee ) 和非对映选择性(高达 >20:1 dr)。








































 京公网安备 11010802027423号
京公网安备 11010802027423号