当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Tetrazolo[1,5-a]pyridine-Containing π-Conjugated Systems: Synthesis, Properties, and Semiconducting Characteristics
Organic Letters ( IF 4.9 ) Pub Date : 2022-05-23 , DOI: 10.1021/acs.orglett.2c01310 Shunsuke Tamba 1 , Masashi Nitani 1 , Takuji Seo 1 , Hajime Nitta 1 , Hikaru Tanaka 2 , Kazutake Hagiya 2 , Yoshio Aso 1 , Yutaka Ie 1, 3
Organic Letters ( IF 4.9 ) Pub Date : 2022-05-23 , DOI: 10.1021/acs.orglett.2c01310 Shunsuke Tamba 1 , Masashi Nitani 1 , Takuji Seo 1 , Hajime Nitta 1 , Hikaru Tanaka 2 , Kazutake Hagiya 2 , Yoshio Aso 1 , Yutaka Ie 1, 3
Affiliation
|
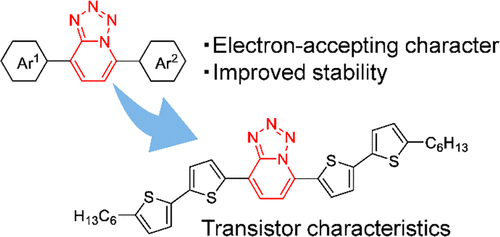
The incorporation of an electron-accepting unit into π-conjugated systems is an important approach to modulate the physical properties of such molecules. To investigate the potential of tetrazolo[1,5-a]pyridine as an electron-accepting unit, a series of diarylated tetrazolo[1,5-a]pyridine derivatives was synthesized by treating the corresponding diarylated pyridine N-oxide with diphenylphosphoryl azide. Thermogravimetric analyses of these molecules indicated that they possessed good thermal stability. The bithiophene-substituted tetrazolo[1,5-a]pyridine compound showed stable transistor characteristics under repeated bias conditions.
中文翻译:

含有四唑并[1,5-a]吡啶的π共轭体系:合成、性质和半导体特性
将电子接受单元结合到 π 共轭体系中是调节此类分子物理性质的重要方法。为了研究四唑并[1,5 - a ]吡啶作为电子接受单元的潜力,通过用二苯基磷酰叠氮化物处理相应的二芳基化吡啶N-氧化物,合成了一系列二芳基化四唑并[1,5- a ]吡啶衍生物。这些分子的热重分析表明它们具有良好的热稳定性。噻吩取代的四唑并[1,5- a ]吡啶化合物在重复偏压条件下表现出稳定的晶体管特性。
更新日期:2022-05-23
中文翻译:

含有四唑并[1,5-a]吡啶的π共轭体系:合成、性质和半导体特性
将电子接受单元结合到 π 共轭体系中是调节此类分子物理性质的重要方法。为了研究四唑并[1,5 - a ]吡啶作为电子接受单元的潜力,通过用二苯基磷酰叠氮化物处理相应的二芳基化吡啶N-氧化物,合成了一系列二芳基化四唑并[1,5- a ]吡啶衍生物。这些分子的热重分析表明它们具有良好的热稳定性。噻吩取代的四唑并[1,5- a ]吡啶化合物在重复偏压条件下表现出稳定的晶体管特性。







































 京公网安备 11010802027423号
京公网安备 11010802027423号