当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
From Serendipity to Rational Design: Heteroleptic Dirhodium Amidate Complexes for Diastereodivergent Asymmetric Cyclopropanation
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-04-14 , DOI: 10.1021/jacs.2c02258 Fabio Pasquale Caló 1 , Anne Zimmer 1 , Giovanni Bistoni 1 , Alois Fürstner 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-04-14 , DOI: 10.1021/jacs.2c02258 Fabio Pasquale Caló 1 , Anne Zimmer 1 , Giovanni Bistoni 1 , Alois Fürstner 1
Affiliation
|
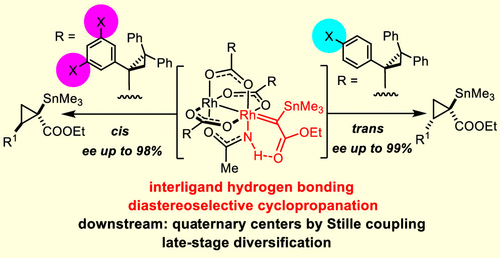
A heteroleptic dirhodium paddlewheel complex comprising three chiral carboxylate ligands and one achiral acetamidate ligand has recently been found to be uniquely effective in catalyzing the asymmetric cyclopropanation of olefins with α-stannylated (silylated and germylated) α-diazoacetate derivatives. A number of control experiments in combination with detailed computational studies provide compelling evidence that an interligand hydrogen bond between the −NH group of the amidate and the ester carbonyl group of the reactive rhodium carbene intermediate plays a quintessential role in the stereodetermining transition state. The penalty for distorting this array outweighs steric arguments and renders two of the four conceivable transitions states unviable. Based on this mechanistic insight, the design of the parent catalyst is revisited herein: placement of appropriate peripheral substituents allows high levels of diastereocontrol to be imposed upon cyclopropanation, which the original catalyst lacks. Because the new complexes allow either trans- or cis-configured stannylated cyclopropanes to be made selectively and in excellent optical purity, this transformation also marks a rare case of diastereodivergent asymmetric catalysis. The products are amenable to stereospecific cross coupling with aryl halides or alkenyl triflates; these transformations appear to be the first examples of the formation of stereogenic quaternary carbon centers by the Stille reaction; carbonylative coupling is also achieved. Moreover, tin/lithium exchange affords chiral lithium enolates, which can be intercepted with a variety of electrophilic partners. The virtues and inherent flexibility of this new methodology are illustrated by an efficient synthesis of two salinilactones, extremely scarce bacterial metabolites with signaling function involved in the self-regulatory growth inhibition of the producing strain.
中文翻译:

从巧合到理性设计:用于非对映异构不对称环丙烷化的异配氨基二铑配合物
最近发现包含三个手性羧酸盐配体和一个非手性乙酰胺酸盐配体的杂配二铑桨轮配合物在催化烯烃与 α-甲硅烷基化(甲硅烷基化和锗酰化)α-重氮乙酸酯衍生物的不对称环丙烷化方面具有独特的效果。许多对照实验与详细的计算研究相结合,提供了令人信服的证据,表明酰胺化物的 -NH 基团和反应性铑卡宾中间体的酯羰基之间的配体间氢键在立体决定过渡态中起典型作用。扭曲这个数组的代价超过了空间参数,并使四个可能的转换状态中的两个不可行。基于这种机理洞察力,本文重新审视了母体催化剂的设计:适当的外围取代基的放置允许对环丙烷化施加高水平的非对映控制,这是原始催化剂所缺乏的。由于新的配合物允许选择性地制备反式或顺式构型的亚烷基环丙烷并具有出色的光学纯度,因此这种转变也标志着非对映发散不对称催化的罕见情况。该产品可与芳基卤化物或烯基三氟甲磺酸酯进行立体定向交叉偶联;这些转化似乎是通过 Stille 反应形成立体季碳中心的第一个例子;也实现了羰基化偶联。此外,锡/锂交换提供手性锂烯醇化物,它可以被多种亲电子伙伴拦截。
更新日期:2022-04-14
中文翻译:

从巧合到理性设计:用于非对映异构不对称环丙烷化的异配氨基二铑配合物
最近发现包含三个手性羧酸盐配体和一个非手性乙酰胺酸盐配体的杂配二铑桨轮配合物在催化烯烃与 α-甲硅烷基化(甲硅烷基化和锗酰化)α-重氮乙酸酯衍生物的不对称环丙烷化方面具有独特的效果。许多对照实验与详细的计算研究相结合,提供了令人信服的证据,表明酰胺化物的 -NH 基团和反应性铑卡宾中间体的酯羰基之间的配体间氢键在立体决定过渡态中起典型作用。扭曲这个数组的代价超过了空间参数,并使四个可能的转换状态中的两个不可行。基于这种机理洞察力,本文重新审视了母体催化剂的设计:适当的外围取代基的放置允许对环丙烷化施加高水平的非对映控制,这是原始催化剂所缺乏的。由于新的配合物允许选择性地制备反式或顺式构型的亚烷基环丙烷并具有出色的光学纯度,因此这种转变也标志着非对映发散不对称催化的罕见情况。该产品可与芳基卤化物或烯基三氟甲磺酸酯进行立体定向交叉偶联;这些转化似乎是通过 Stille 反应形成立体季碳中心的第一个例子;也实现了羰基化偶联。此外,锡/锂交换提供手性锂烯醇化物,它可以被多种亲电子伙伴拦截。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号