Journal of Fluorine Chemistry ( IF 1.7 ) Pub Date : 2021-12-05 , DOI: 10.1016/j.jfluchem.2021.109931 Gang Yang 1 , Chengping Zhang 1 , Hengdao Quan 2
|
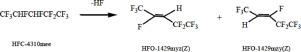
Studies were conducted on the vapor-phase dehydrofluorination of 2H,3H-decafluoropentane. It was found that 2H,3H-decafluoropentane could be dehydrofluorinated to give Z-1,1,1,2,4,4,5,5,5-nonafluoro-2-pentene and Z-1,1,1,3,4,4,5,5,5-nonafluoro-2-pentene over chromium fluoride or aluminum fluoride catalysts. With stoichiometric calculation, Z-isomer was the main product in the dehydrofluorination owing to its lower relative energy, which was consistent with the experimental result. Consequently, the reaction mechanisms of selective dehydrofluorination of 2H, 3H-decafluoropentane were proposed.
中文翻译:

2H,3H-十氟戊烷脱氟化氢的研究
对 2H,3H-十氟戊烷的气相脱氟化氢进行了研究。发现 2H,3H-十氟戊烷可以脱氟化氢得到 Z-1,1,1,2,4,4,5,5,5-nonafluoro-2-pentene 和 Z-1,1,1,3, 4,4,5,5,5-nonafluoro-2-pentene 在氟化铬或氟化铝催化剂上。通过化学计量计算,Z-异构体由于其相对能量较低,是脱氟化氢反应的主要产物,这与实验结果一致。因此,提出了2H、3H-十氟戊烷选择性脱氟化氢的反应机理。






































 京公网安备 11010802027423号
京公网安备 11010802027423号