当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
New approaches to ondansetron and alosetron inspire a versatile, flow photochemical method for indole synthesis
Chemical Communications ( IF 4.3 ) Pub Date : 2021-11-26 , DOI: 10.1039/d1cc05700f Wei Sun 1 , William A T Raimbach 1 , Luke D Elliott 2 , Kevin I Booker-Milburn 2 , David C Harrowven 1
Chemical Communications ( IF 4.3 ) Pub Date : 2021-11-26 , DOI: 10.1039/d1cc05700f Wei Sun 1 , William A T Raimbach 1 , Luke D Elliott 2 , Kevin I Booker-Milburn 2 , David C Harrowven 1
Affiliation
|
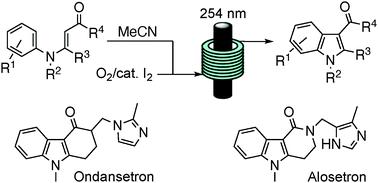
An oxidative photocyclisation of N-arylenaminones to indoles is described, that mirrors the Fischer indole synthesis but uses anilines in place of arylhydrazines. Its value is exemplified with new approaches to the WHO-listed APIs ondansetron and alosetron.
中文翻译:

昂丹司琼和阿洛司琼的新方法激发了吲哚合成的多功能流动光化学方法
描述了N-亚芳基氨基酮氧化光环化为吲哚,这反映了 Fischer 吲哚合成,但使用苯胺代替芳基肼。它的价值体现在对 WHO 列出的 API 昂丹司琼和阿洛司琼的新方法中。
更新日期:2021-12-04
中文翻译:

昂丹司琼和阿洛司琼的新方法激发了吲哚合成的多功能流动光化学方法
描述了N-亚芳基氨基酮氧化光环化为吲哚,这反映了 Fischer 吲哚合成,但使用苯胺代替芳基肼。它的价值体现在对 WHO 列出的 API 昂丹司琼和阿洛司琼的新方法中。















































 京公网安备 11010802027423号
京公网安备 11010802027423号