European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2021-11-02 , DOI: 10.1016/j.ejmech.2021.113965 Yu-Hang Yan 1 , Wenfang Li 2 , Wei Chen 3 , Chao Li 3 , Kai-Rong Zhu 1 , Ji Deng 1 , Qing-Qing Dai 1 , Ling-Ling Yang 3 , Zhenling Wang 2 , Guo-Bo Li 1
|
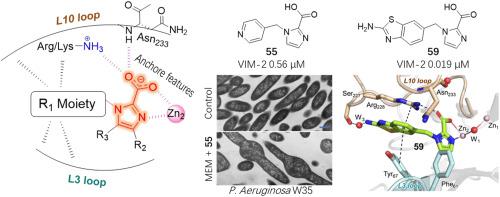
Production of metallo-β-lactamases (MBLs) in bacterial pathogens is an important cause of resistance to the ‘last-resort’ carbapenem antibiotics. Development of effective MBL inhibitors to reverse carbapenem resistance in Gram-negative bacteria is still needed. We herein report X-ray structure-guided optimization of 1H-imidazole-2-carboxylic acid (ICA) derivatives by considering how to engage with the active-site flexible loops and improve penetration into Gram-negative bacteria. Structure-activity relationship studies revealed the importance of appropriate substituents at ICA 1-position to achieve potent inhibition to class B1 MBLs, particularly the Verona Integron-encoded MBLs (VIMs), mainly by involving ingenious interactions with the flexible active site loops as observed by crystallographic analyses. Of the tested ICA inhibitors, 55 displayed most potent synergistic antibacterial activity with meropenem against engineered Escherichia coli strains and even intractable clinically isolated Pseudomonas aeruginosa producing VIM-2 MBL. The morphologic and internal structural changes of bacterial cells after treatment further demonstrated that 55 crossed the outer membrane and reversed the activity of meropenem. Moreover, 55 showed good pharmacokinetic and safety profile in vivo, which could be a potential candidate for combating VIM-mediated Gram-negative carbapenem resistance.
中文翻译:

1H-咪唑-2-羧酸衍生物的结构引导优化,可提供有效的 VIM 型金属-β-内酰胺酶抑制剂
细菌病原体中金属-β-内酰胺酶 (MBL) 的产生是对“最后手段”碳青霉烯类抗生素产生耐药性的重要原因。仍然需要开发有效的 MBL 抑制剂来逆转革兰氏阴性菌对碳青霉烯类的耐药性。我们在此报告了 1 H的 X 射线结构引导优化- 咪唑-2-羧酸(ICA)衍生物,通过考虑如何与活性位点柔性环结合并提高对革兰氏阴性细菌的渗透。构效关系研究揭示了在 ICA 1 位适当的取代基对于实现对 B1 类 MBLs,特别是 Verona Integron 编码的 MBLs (VIMs) 的有效抑制的重要性,主要是通过与灵活的活性位点环的巧妙相互作用,如观察到的那样晶体学分析。在测试的 ICA 抑制剂中,55种与美罗培南对工程大肠杆菌菌株甚至难治的临床分离的铜绿假单胞菌具有最强的协同抗菌活性生产 VIM-2 MBL。处理后细菌细胞的形态和内部结构变化进一步证明55穿过外膜并逆转美罗培南的活性。此外,55在体内显示出良好的药代动力学和安全性,这可能是对抗 VIM 介导的革兰氏阴性碳青霉烯类耐药性的潜在候选者。






























 京公网安备 11010802027423号
京公网安备 11010802027423号