当前位置:
X-MOL 学术
›
J. Phys. Chem. C
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Lithium Ion Disorder and Conduction Mechanism in LiCe(BH4)3Cl
The Journal of Physical Chemistry C ( IF 3.3 ) Pub Date : 2016-08-16 00:00:00 , DOI: 10.1021/acs.jpcc.6b06564
Young-Su Lee 1 , Morten B. Ley 2, 3 , Torben R. Jensen 2 , Young Whan Cho 1
The Journal of Physical Chemistry C ( IF 3.3 ) Pub Date : 2016-08-16 00:00:00 , DOI: 10.1021/acs.jpcc.6b06564
Young-Su Lee 1 , Morten B. Ley 2, 3 , Torben R. Jensen 2 , Young Whan Cho 1
Affiliation
|
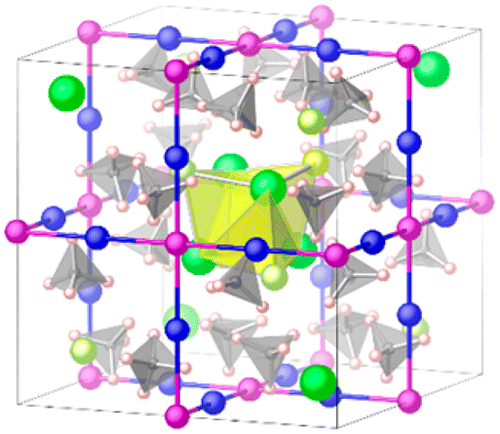
We investigate the diffusion mechanism of Li ions in LiCe(BH4)3Cl, which exhibits fast Li ion conduction. It was previously shown that eight Li ions partially occupy the 12d Wyckoff sites in the I4̅3m structure and the Li ion diffusion takes place via jumping through the three-dimensional network of the 12d sites. In this study, we employ first-principles nudged elastic band simulation to elucidate the diffusion mechanism and discover that the Li ion does not directly jump to the neighboring 12d site, but instead passes through the closest 6b site. Moreover, the 6b site turns out to be another stable Li ion site, not just a transient point during a jump event. The occupation of the 6b site and the Li ion diffusion mechanism were assured by first-principles molecular dynamics simulations. The partial occupancy of the 12d site and 6b site at 500 K is approximately 1/2 and 1/3, respectively. The experimental diffraction data can be consistently interpreted. The peculiar crystal structure of LiCe(BH4)3Cl allowing efficient and fast Li ion diffusion is again highlighted together with the role of [BH4]− ion in thermodynamically stabilizing LiCe(BH4)3Cl.
中文翻译:

LiCe(BH 4)3 Cl中的锂离子无序与导电机理
我们研究了Li离子在LiCe(BH 4)3 Cl中的扩散机理,该机理表现出快速的Li离子传导性。之前显示,8个Li离子部分地占据的12 d在Wyckoff的站点我43米结构和Li离子扩散穿过12个的三维网络通过跳跃发生d部位。在这项研究中,我们采用第一性原理微动松紧带模拟来阐明扩散机理,并发现锂离子不会直接跳到相邻的12 d位置,而是穿过最近的6 b位置。此外,6 b该位点被证明是另一个稳定的锂离子位点,而不仅仅是跳跃事件中的一个过渡点。第一性原理分子动力学模拟确保了6b位的占据和Li离子扩散机制。在500 K处12 d和6 b的部分占用分别约为1/2和1/3。实验衍射数据可以得到一致的解释。LiCe(BH 4)3 Cl的独特晶体结构允许有效和快速的Li离子扩散,再加上[BH 4 ] -离子在热力学稳定LiCe(BH 4)3 Cl中的作用,再次得到强调。
更新日期:2016-08-16
中文翻译:

LiCe(BH 4)3 Cl中的锂离子无序与导电机理
我们研究了Li离子在LiCe(BH 4)3 Cl中的扩散机理,该机理表现出快速的Li离子传导性。之前显示,8个Li离子部分地占据的12 d在Wyckoff的站点我43米结构和Li离子扩散穿过12个的三维网络通过跳跃发生d部位。在这项研究中,我们采用第一性原理微动松紧带模拟来阐明扩散机理,并发现锂离子不会直接跳到相邻的12 d位置,而是穿过最近的6 b位置。此外,6 b该位点被证明是另一个稳定的锂离子位点,而不仅仅是跳跃事件中的一个过渡点。第一性原理分子动力学模拟确保了6b位的占据和Li离子扩散机制。在500 K处12 d和6 b的部分占用分别约为1/2和1/3。实验衍射数据可以得到一致的解释。LiCe(BH 4)3 Cl的独特晶体结构允许有效和快速的Li离子扩散,再加上[BH 4 ] -离子在热力学稳定LiCe(BH 4)3 Cl中的作用,再次得到强调。

































 京公网安备 11010802027423号
京公网安备 11010802027423号