Journal of Molecular Structure ( IF 4.0 ) Pub Date : 2021-08-30 , DOI: 10.1016/j.molstruc.2021.131395 Hong Sun 1, 2, 3 , Liyuan Deng 2, 3 , Weiyin Hu 1, 3 , Tianhui Liao 1, 3 , Weike Liao 4 , Huifang Chai 5 , Chunshen Zhao 1, 2, 3
|
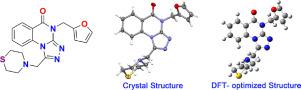
In this present research, the title compound 4-(furan-2-ylmethyl)-1-(thiomorpholinomethyl)-[1,2,4]triazolo[4,3-a]quinazolin-5(4H)-one(1) was synthesized, and its single crystal was produced in acetone. The use of 1H NMR, 13C NMR, FT-IR spectroscopy and MS determined its structure. Using X-ray diffraction, the single crystal of the title compound was measured. At the same time, the optimized molecular crystal structure of this small molecule was determined according to DFT calculations adopting B3LYP/6–311 G (2d, p) basis set. The conformational isomer 1–3 is the most stable conformation and is consistent with the conformation derived from X-ray diffraction. Hirshfeld surface analysis and 2D fingerprint were given to support the quantitative analysis of intermolecular interactions and contacts caused by supramolecular accumulation in crystals.
中文翻译:

4-(furan-2-ylmethyl)-1-(thiomorpholinomethyl)-[1,2,4]triazolo[4,3-a]quinazon-5(4H)-one的合成、晶体结构、振动特性和DFT研究
在本研究中,标题化合物 4-(furan-2-ylmethyl)-1-(thiomorpholinomethyl)-[1,2,4]triazolo[4,3 - a ]quinazolin-5( 4H )-one( 1 )合成,其单晶在丙酮中制得。使用1 H NMR、13 C NMR、FT-IR光谱和MS确定其结构。使用X-射线衍射,测量了标题化合物的单晶。同时,采用B3LYP/6-311 G(2d,p)基组,根据DFT计算确定了该小分子的优化分子晶体结构。构象异构体1–3是最稳定的构象,与 X 射线衍射得到的构象一致。Hirshfeld 表面分析和 2D 指纹用于支持由晶体中超分子积累引起的分子间相互作用和接触的定量分析。







































 京公网安备 11010802027423号
京公网安备 11010802027423号